Sign In to View Organizational & Contract Pricing.
Select a Size
Change View
| Size/SKU | Availability | Price |
|---|
About This Item
Empirical Formula (Hill Notation):
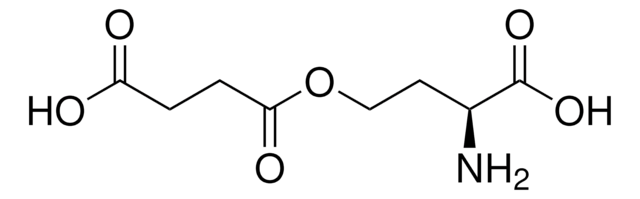
C8H13NO6
CAS Number:
Molecular Weight:
219.19
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.26
MDL number:
Pricing and availability is not currently available.
Product Name
O-Succinyl-L-homoserine,
assay
≥98.0% (TLC)
Quality Segment
form
powder
technique(s)
ligand binding assay: suitable
color
white
storage temp.
−20°C
SMILES string
N[C@@H](CCOC(=O)CCC(O)=O)C(O)=O
InChI
1S/C8H13NO6/c9-5(8(13)14)3-4-15-7(12)2-1-6(10)11/h5H,1-4,9H2,(H,10,11)(H,13,14)/t5-/m0/s1
InChI key
GNISQJGXJIDKDJ-YFKPBYRVSA-N
1 of 1
This Item | |||
|---|---|---|---|
| form powder | form powder | form powder | form powder |
| technique(s) ligand binding assay: suitable | technique(s) ligand binding assay: suitable | technique(s) ligand binding assay: suitable | technique(s) ligand binding assay: suitable |
| assay ≥98.0% (TLC) | assay ≥97.0% (HPLC) | assay ≥98.0% (TLC) | assay >98% (TLC) |
| storage temp. −20°C | storage temp. 2-8°C | storage temp. −20°C | storage temp. −20°C |
| color white | color white to faint beige | color white to off-white | color - |
| Quality Level 100 | Quality Level 100 | Quality Level 100 | Quality Level 200 |
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.