Select a Size
| Size/SKU | Availability | Price |
|---|
About This Item
Product Name
Oxotremorine M, solid
form
solid
Quality Segment
color
white
solubility
H2O: 24 mg/mL, ethanol: 4.6 mg/mL
storage temp.
2-8°C
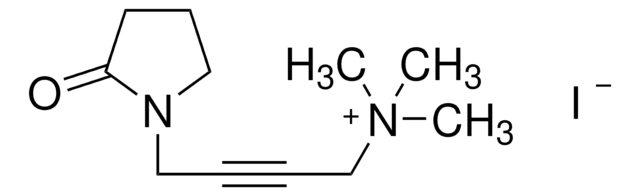
SMILES string
[I-].C[N+](C)(C)CC#CCN1CCCC1=O
InChI
1S/C11H19N2O.HI/c1-13(2,3)10-5-4-8-12-9-6-7-11(12)14;/h6-10H2,1-3H3;1H/q+1;/p-1
InChI key
VVLMSCJCXMBGDI-UHFFFAOYSA-M
Gene Information
human ... CHRM1(1128), CHRM2(1129), CHRM3(1131), CHRM4(1132), CHRM5(1133)
Application
Biochem/physiol Actions
Features and Benefits
1 of 1
This Item | |||
|---|---|---|---|
| form solid | form solid | form powder | form solid |
| Quality Level 200 | Quality Level 100 | Quality Level 100 | Quality Level 100 |
| storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C | storage temp. 2-8°C |
| solubility H2O: 24 mg/mL, ethanol: 4.6 mg/mL | solubility 45% (w/v) aq 2-hydroxypropyl-β-cyclodextrin: 4.3 mg/mL, ethanol: 5.6 mg/mL, H2O: insoluble | solubility H2O: 5 mg/mL, clear (warmed) | solubility DMSO: soluble >5 mg/mL at 60 °C |
| color white | color white | color white to beige | color white |
| Gene Information human ... CHRM1(1128), CHRM2(1129), CHRM3(1131), CHRM4(1132), CHRM5(1133) | Gene Information human ... CHRM3(1131), CHRM4(1132) | Gene Information - | Gene Information rat ... Htr1a(24473) |
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 1 Inhalation - Acute Tox. 2 Dermal - Acute Tox. 2 Oral
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.