E4906
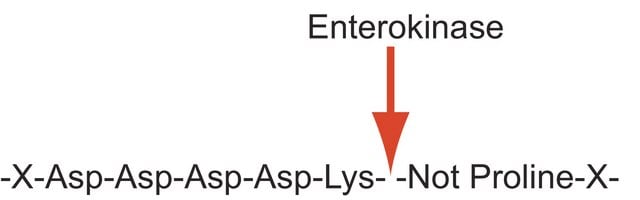
Enterokinase from bovine intestine
BioUltra, recombinant, expressed in E. coli, ≥20 units/mg protein, ≥95% (SDS-PAGE)
Sinónimos:
Enteropeptidase
About This Item
Productos recomendados
recombinant
expressed in E. coli
product line
BioUltra
assay
≥95% (SDS-PAGE)
form
solution
specific activity
≥20 units/mg protein
mol wt
150 kDa
packaging
vial of ~0.2 unit
concentration
≥0.1 mg/mL
shipped in
wet ice
storage temp.
−20°C
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
Adjust the concentration of the fusion protein to 1.5 mg/ml and a pH between 7.0-8.0 with 500 mM Tris-HCl, pH 8.0, 2.0 mM CaCl2, and 1% Tween® 20
Add enterokinase to fusion protein solution at a ratio of ~ 0.02 units per 1 mg fusion protein and mix
Incubate reaction mixture at ~25 °C for 16 hours
Biochem/physiol Actions
Physical properties
Unit Definition
Physical form
Legal Information
Storage Class
10 - Combustible liquids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico