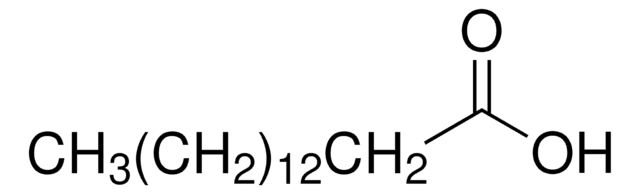PCC300C15C
Pellicon® Capsule with Ultracel® Membrane
C Screen, 1.5 m2 AseptiQuik® G connectors
Sinónimos:
Pellicon® Capsule with Ultracel® Membrane
About This Item
Productos recomendados
material
EPDM rubber seal
PPO/PS blend (core)
PPO/PS blend housing
TPE seal
composite regenerated cellulose (CRC) membrane
epoxy adhesive
polyester screen
polypropylene screen
polyurethane adhesive
Quality Level
sterility
sterile; γ-irradiated
product line
Pellicon®
feature
holdup volume 456 mL (in feed channel)
holdup volume 741 mL (in permeate channel)
packaging
package of 1
parameter
max. inlet pressure at 75 psi (5.1 bar) and 4-30 °C
max. transmembrane pressure at 45 psi (3.1 bar) and 4-30 °C
4-6 L/min-m2 flow rate
technique(s)
ultrafiltration: suitable
diam.
10.3 cm (4.1 in.)
filtration area
1.5 m2
screen size
, Type C screen (coarse screen)
size
43.7 cm (17.2 in.) , Length
impurities
≤5 ppm Meets TOC (in retentate effluent, after a WFI flush of 20 L/m²)
matrix
Ultracel®
pore size
300 kDa
pH range
2-13
storage temp.
15-25°C
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Permeate: AseptiQuik® G Connector
Application
Features and Benefits
- Plug ′n play, holderless design - easy to install, safe to remove
- Gamma Sterilized and preseravtive-free - ready to process in minutes
- True single-use, self-contained capusle - fast, safe, and flexible batch turnaround
- Proven Ultracel® membrane and C screen - high recovery, superior mass transfer, solvent resistance
- Pellicon® TFF proven performance - true linear scalability within Pellicon® TFF families
Packaging
Other Notes
- Comprehensive documentation to support qualification, risk assessment and process optimization needs.
- Consolidation of product specific testing, quality and regulatory information to simplify compliance requirements.
- Convenient 24/7 access to up-to-date product information.
Legal Information
Not finding the right product?
Try our Herramienta de selección de productos.
Related product
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Artículos
Our overview of scalable tangential flow filtration for viral vector production will provide key considerations for developing and optimizing your downstream process.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico