593095
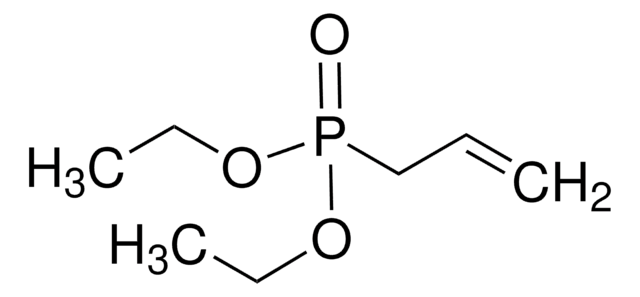
Diethyl (2-methylallyl)phosphonate
97%
About This Item
Productos recomendados
Ensayo
97%
idoneidad de la reacción
reaction type: C-C Bond Formation
índice de refracción
n20/D 1.4380 (lit.)
bp
62 °C/0.1 mmHg (lit.)
densidad
1.013 g/mL at 25 °C (lit.)
grupo funcional
phosphonate
cadena SMILES
CCOP(=O)(CC(C)=C)OCC
InChI
1S/C8H17O3P/c1-5-10-12(9,11-6-2)7-8(3)4/h3,5-7H2,1-2,4H3
Clave InChI
QOZGSMHGXZMADD-UHFFFAOYSA-N
Descripción general
Aplicación
It can also be used as a reactant for:
- Enantioselective total synthesis of 10-isocyano-4-cadinene as antifouling agent.[4]
- Regiospecific preparation of 4-oxo-2-alkenylphosphonates (OAP) via silylation followed by Friedel-Crafts acylation and isomerization. OAP can serve as building blocks for the construction of polyethylenic chains.[5]
- The synthesis of azaphosphone as a potent analgesic/anti-inflammatory agents.[2]
- Enantioselective synthesis of 10-isocyano-4-cadinene and its stereoisomers with antifouling activity
- Preparation of 4-Oxo-2-alkenylphosphonates via silylation followed by regiospecific Friedel-Crafts acylation and isomerization
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2
Código de clase de almacenamiento
10 - Combustible liquids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
>230.0 °F - closed cup
Punto de inflamabilidad (°C)
> 110 °C - closed cup
Equipo de protección personal
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Active Filters
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico