M29800
2-Methylbenzimidazole
98%
Synonym(s):
2-Methyl-1H-1,3-benzodiazole, 2-Methyl-1H-benzimidazole, 2-Methyl-1H-benzo[d]imidazole, 2-Methylbenzoimidazole
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C8H8N2
CAS Number:
Molecular Weight:
132.16
Beilstein:
112264
EC Number:
MDL number:
UNSPSC Code:
12352100
eCl@ss:
32151902
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
mp
175-177 °C (lit.)
SMILES string
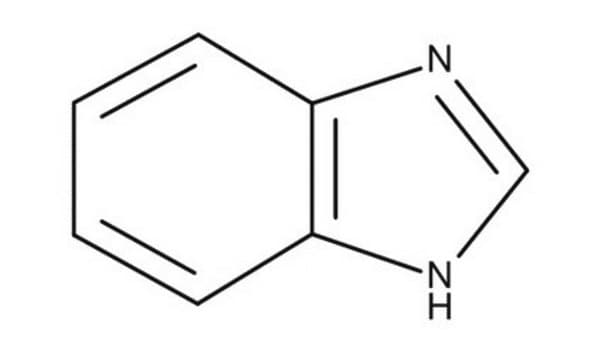
Cc1nc2ccccc2[nH]1
InChI
1S/C8H8N2/c1-6-9-7-4-2-3-5-8(7)10-6/h2-5H,1H3,(H,9,10)
InChI key
LDZYRENCLPUXAX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- 2-Methylbenzimidazole is an important pharmacophore widely used in medicinal chemistry for the synthesis of various antibacterial and antifungal agents.
- It can be used as a key precursor to synthesize substituted benzimidazo[1,2-a]quinolones.
- It can be used in the synthesis of reversible solid-to-liquid phase transition coordination polymer crystals.
- 2-Methylbenzimidazole also exhibits corrosion inhibition.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Reversible solid-to-liquid phase transition of coordination polymer crystals.
Umeyama D, et al.
Journal of the American Chemical Society, 137(2), 864-870 (2015)
Deidré van den Berg et al.
Bioorganic & medicinal chemistry, 15(11), 3692-3702 (2007-04-10)
We have recently reported that a series of (E)-8-styrylcaffeines and (E)-2-styrylbenzimidazoles are moderate to very potent competitive inhibitors of monoamine oxidase B (MAO-B). The most potent member of the series was found to be (E)-8-(3-chlorostyryl)caffeine (CSC) with an enzyme-inhibitor dissociation
Synthesis and biological evaluation of substituted benzimidazoles.
Shah K, et al.
Journal of the Indian Chemical Society, 93, 1009-1018 (2016)
Theoretical study of benzimidazole and its derivatives and their potential activity as corrosion inhibitors.
Obot I B and Obi-Egbedi N O
Corrosion Science, 52(2), 657-660 (2010)
Novel strategy for synthesis of substituted benzimidazo [1, 2-a] quinolines.
Kato J Y, et al.
Organic Letters, 15(14), 3794-3797 (2013)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service