796883
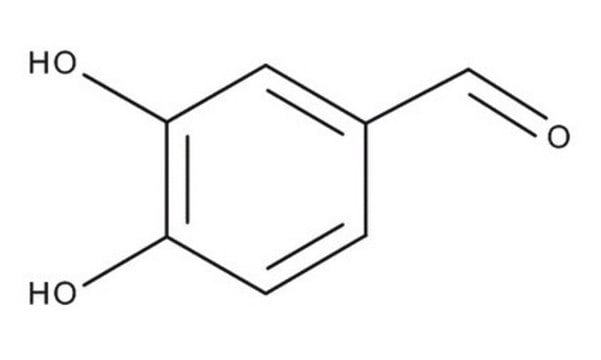
3,4-dihydroxyphenylacetone
95%
Synonym(s):
1-(3,4-Dihydroxyphenyl)-2-propanone, 1-(3′,4′-Dihydroxyphenyl)-2-propanone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C9H10O3
CAS Number:
Molecular Weight:
166.17
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
95%
form
powder
functional group
ketone
SMILES string
CC(CC1=CC(O)=C(O)C=C1)=O
InChI
1S/C9H10O3/c1-6(10)4-7-2-3-8(11)9(12)5-7/h2-3,5,11-12H,4H2,1H3
InChI key
JQXBETDGCMQLMK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
3,4-dihydroxyphenylacetoneis an alkylbenzene. 3,4-dihydroxyphenylacetone is the product of oxidative deamination of α-methylDopa. The product is intended for forensic and research purposes.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Mammalian Dopa decarboxylase: structure, catalytic activity and inhibition
Bertoldi M.
Archives of Biochemistry and Biophysics, 546, 1-7 (2014)
Mohammadreza Shariatgorji et al.
Nature methods, 16(10), 1021-1028 (2019-09-25)
We present a mass spectrometry imaging (MSI) approach for the comprehensive mapping of neurotransmitter networks in specific brain regions. Our fluoromethylpyridinium-based reactive matrices facilitate the covalent charge-tagging of molecules containing phenolic hydroxyl and/or primary or secondary amine groups, including dopaminergic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service