348015
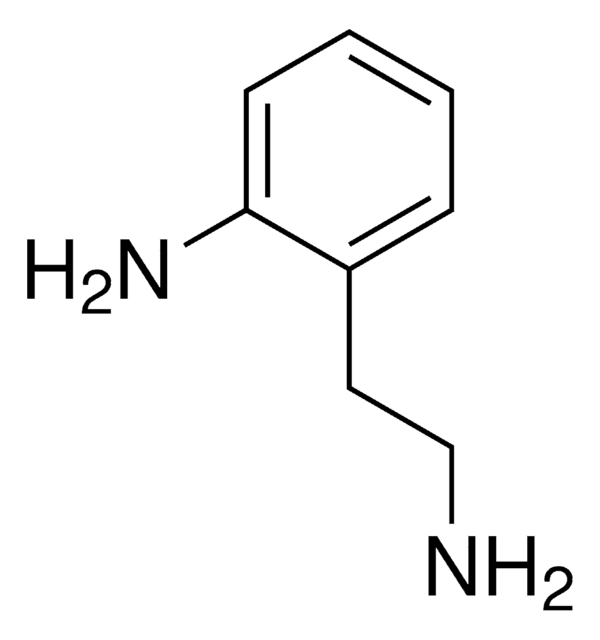
2-Aminobenzylamine
98%
Synonym(s):
(2-Aminomethylphenyl)amine, 2-(Aminomethyl)aniline, 2-(Aminomethyl)benzenamine, 2-Amino-1-benzylamine, [(2-Aminophenyl)methyl]amine, o-Aminobenzylamine
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
H2NC6H4CH2NH2
CAS Number:
Molecular Weight:
122.17
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
solid
mp
58-61 °C (lit.)
functional group
amine
SMILES string
NCc1ccccc1N
InChI
1S/C7H10N2/c8-5-6-3-1-2-4-7(6)9/h1-4H,5,8-9H2
InChI key
GVOYKJPMUUJXBS-UHFFFAOYSA-N
General description
2-Aminobenzylamine undergoes three-component cyclisation reactions with methyl 3,3,3-trifluoropyruvate, 2-aminobenzylamine and oxo compounds to afford regio- and stereoisomers of tetrahydropyrroloquinazolinones.
Application
2-Aminobenzylamine may be used:
- in the synthesis of 1,2,3,4-tetrahydroquinazoline oxime, via condensation reaction with 2-(naphthalen-2-yl)-2-oxoacetaldehyde oxime
- in the synthesis of alkyl 5H-1,4-benzodiazepine-3-carboxylates
- to modify the phosphate groups on phosphoserine peptides
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
G Sirikçi et al.
Acta chimica Slovenica, 59(4), 904-911 (2013-09-26)
A novel 1,2,3,4-tetrahydroquinazoline oxime was synthesised from a condensation reaction of 2-(naphthalen-2-yl)-2-oxoacetaldehyde oxime with 2-aminobenzylamine. Subsequently, a-imine oxime complexes of this compound that formed with Co(III) and Ni(II) metal ions were obtained. All structures were characterised by spectral methods (FT-IR
Orazio A Attanasi et al.
The Journal of organic chemistry, 76(20), 8320-8328 (2011-09-13)
A novel and simple one-pot synthesis of 3-substituted 2,5,6,7-tetrahydro-1H-1,4-diazepin-2-ones from 1,2-diaza-1,3-dienes (DDs) and N-unsubstituted aliphatic 1,3-diamines is described. Here we also report a procedure to selectively obtain alkyl 5H-1,4-benzodiazepine-3-carboxylates from the DDs and 2-aminobenzylamine. Both processes occur by means of
Wen-Yun Hsueh et al.
Journal of medicinal chemistry, 64(3), 1435-1453 (2021-01-26)
In this paper, we present a copper(I)-catalyzed nitrile-addition/N-arylation ring-closure cascade for the synthesis of 5,11-dihydro-6H-indolo[3,2-c]quinolin-6-ones from 2-(2-bromophenyl)-N-(2-cyanophenyl)acetamides. Using CuBr and t-BuONa in dimethylformamide (DMF) as the optimal reaction conditions, the cascade reaction gave the target products, in high yields, with
Bohumil Dolenský et al.
Magnetic resonance in chemistry : MRC, 48(5), 375-385 (2010-03-20)
A new three-component cyclisation reactions of methyl 3,3,3-trifluoropyruvate, 2-aminobenzylamine and oxo compounds afforded tetrahydropyrroloquinazolinones of the types 4 and 5 as mixtures of regio- and stereoisomers. Whereas standard 1D NMR spectroscopy was used for a facile assignment of the cyclization
Lukas K Filak et al.
Organometallics, 32(3), 903-914 (2013-02-23)
Six novel ruthenium(II)- and osmium(II)-arene complexes with three modified indolo[3,2-c]quinolines have been synthesized in situ starting from 2-aminoindoloquinolines and 2-pyridinecarboxaldehyde in the presence of [M(p-cymene)Cl(2)](2) (M = Ru, Os) in ethanol. All complexes have been characterized by elemental analysis, spectroscopic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service