326372
Ammonium chloride
99.99% trace metals basis
Synonym(s):
Salmiac
About This Item
Recommended Products
vapor density
1.9 (vs air)
Quality Level
vapor pressure
1 mmHg ( 160.4 °C)
Assay
99.99% trace metals basis
form
crystalline
impurities
≤150.0 ppm Trace Metal Analysis
mp
340 °C (subl.) (lit.)
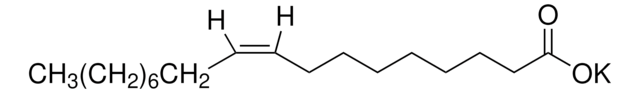
SMILES string
N.Cl
InChI
1S/ClH.H3N/h1H;1H3
InChI key
NLXLAEXVIDQMFP-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
The aldol condensation reaction is an organic reaction introduced by Charles Wurtz, who first prepared the β-hydroxy aldehyde from acetaldehdye in 1872.
The aldol condensation reaction is an organic reaction introduced by Charles Wurtz, who first prepared the β-hydroxy aldehyde from acetaldehdye in 1872.
The aldol condensation reaction is an organic reaction introduced by Charles Wurtz, who first prepared the β-hydroxy aldehyde from acetaldehdye in 1872.
The aldol condensation reaction is an organic reaction introduced by Charles Wurtz, who first prepared the β-hydroxy aldehyde from acetaldehdye in 1872.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service