31460
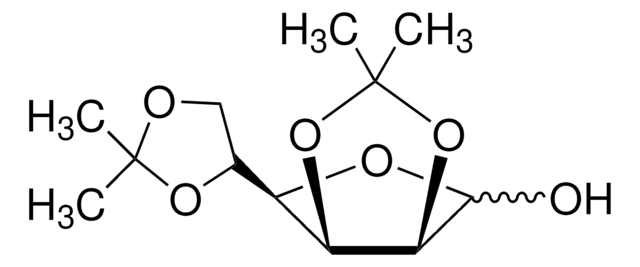
1,2:5,6-Di-O-isopropylidene-α-D-glucofuranose
purum, ≥98.0% (TLC)
Synonym(s):
D-Glucose diacetonide, Diacetone-D-glucose
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C12H20O6
CAS Number:
Molecular Weight:
260.28
Beilstein:
84386
EC Number:
MDL number:
UNSPSC Code:
12352201
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
grade
purum
Quality Level
Assay
≥98.0% (TLC)
optical activity
[α]20/D −11.5±1°, c = 5% in ethanol
mp
110-111 °C (lit.)
SMILES string
CC1(C)OC[C@@H](O1)[C@H]2O[C@@H]3OC(C)(C)O[C@@H]3[C@H]2O
InChI
1S/C12H20O6/c1-11(2)14-5-6(16-11)8-7(13)9-10(15-8)18-12(3,4)17-9/h6-10,13H,5H2,1-4H3/t6-,7+,8-,9-,10-/m1/s1
InChI key
KEJGAYKWRDILTF-JDDHQFAOSA-N
Looking for similar products? Visit Product Comparison Guide
Other Notes
Important protected derivative of glucose; the 3-OH group can be directly manipulated, the 5,6-O-isopropylidene protection is selectively cleavable; oxidation and reduction of the 3-OH leads to an allofuranose derivative; review
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
S. Iacono et al.
Organic Syntheses, 64, 57-57 (1986)
Yoon-Suk Kang et al.
Infection and immunity, 87(8) (2019-06-05)
Brucella is an intracellular bacterial pathogen that causes chronic systemic infection in domesticated livestock and poses a zoonotic infectious risk to humans. The virulence of Brucella is critically dependent on its ability to replicate and survive within host macrophages. Brucella
A. Vasella et al.
Modern Synthetic Methods, 2, 173-173 (1980)
Z Huang et al.
Methods in molecular biology (Clifton, N.J.), 20, 315-353 (1993-01-01)
Two sets of experimental protocols are given for the synthesis of 3',5'-bis-homodeoxyribonucleosides, building blocks for the synthesis of oligodeoxynucleotide analogs where the -O-PO2-O- groups are replaced by -CH2-S-CH2-, -CH2-SO-CH2-, and -CH2-SO2-CH2- units. Conditions are presented for joining these building blocks
E White et al.
Biomedical mass spectrometry, 9(9), 395-405 (1982-09-01)
Two isotope dilution mass spectrometric methods have been developed for the determination of D-glucose in human serum. Each uses a uniformly labeled (13C)glucose as the internal standard. The first method involves conversion of glucose into 1,2:5,6-di-O-isopropylidene-alpha-D-glucofuranose and an extensive clean-up
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![(3AR,5S,6S,6AR)-5-((R)-2,2-DIMETHYL-1,3-DIOXOLAN-4-YL)-2,2-DIMETHYLTETRAHYDROFURO[3,2-D][1,3]DIOXOL-6-OL AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/241/825/1c695a85-5c36-42d3-806a-30876a4dabac/640/1c695a85-5c36-42d3-806a-30876a4dabac.png)