471933
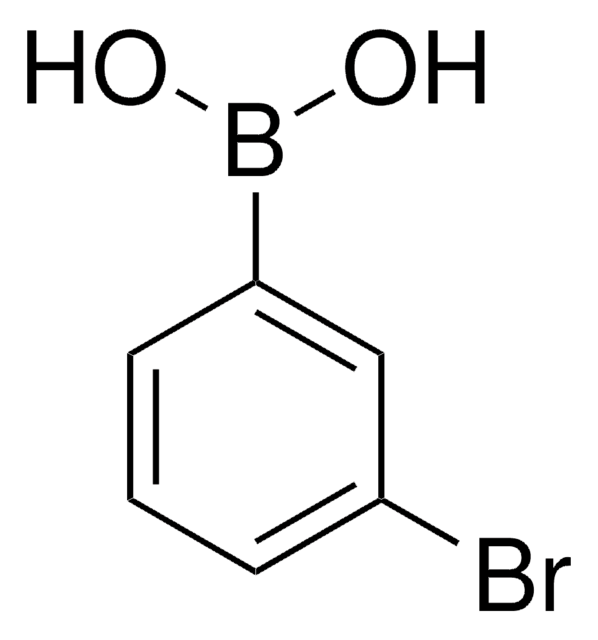
4-Iodophenylboronic acid
≥95.0%
Synonym(s):
B-(4-iodophenyl)-boronic acid, p-Iodophenylboronic acid, p-iodo-benzeneboronic acid
About This Item
Recommended Products
Assay
≥95.0%
mp
326-330 °C (lit.)
SMILES string
OB(O)c1ccc(I)cc1
InChI
1S/C6H6BIO2/c8-6-3-1-5(2-4-6)7(9)10/h1-4,9-10H
InChI key
PELJYVULHLKXFF-UHFFFAOYSA-N
Application
- Copper-mediated ligandless aerobic fluoroalkylation
- Palladium-catalyzed aerobic oxidative cross-coupling reactions
- Recyclable magnetic-nanoparticle-supported palladium catalyst for the Suzuki coupling reactions
- Oxidative hydroxylation using a copper (Cu) catalyst
- Ligand-free palladium-catalyzed Suzuki-Miyaura cross-coupling
- Homocoupling using gold salts as a catalyst
- Ruthenium (Ru)-catalyzed cross-coupling
- CuI-catalyzed Suzuki coupling reactions
- Palladium-catalyzed domino Heck-Mizoroki/Suzuki-Miyaura reactions
- Manganese triacetate-mediated radical additions of arylboronic acids to alkenes
Reagent used in Preparation of
- Pleuromutilin derivatives for ribosomal binding and antibacterial activity via "Click Chemistry"
- Liquid crystalline polyacetylene derivatives
Other Notes
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service