All Photos(1)
About This Item
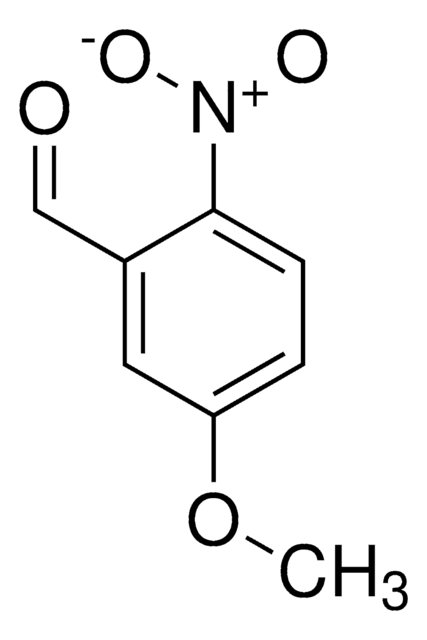
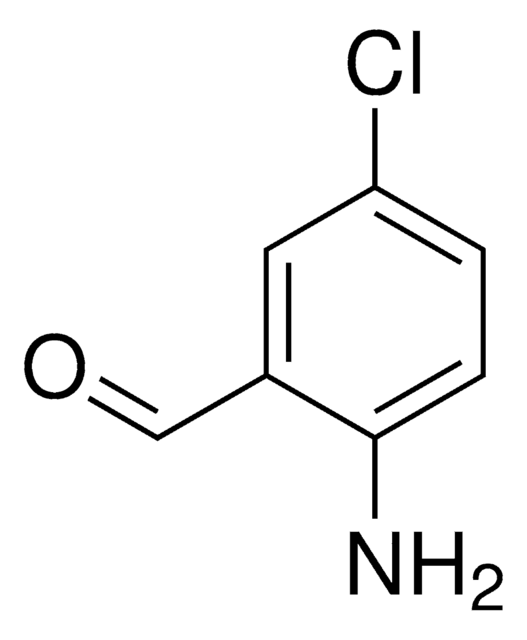
Linear Formula:
ClC6H3(NO2)CHO
CAS Number:
Molecular Weight:
185.56
Beilstein:
743764
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
mp
75-77 °C (lit.)
functional group
aldehyde
chloro
nitro
SMILES string
[H]C(=O)c1cc(ccc1Cl)[N+]([O-])=O
InChI
1S/C7H4ClNO3/c8-7-2-1-6(9(11)12)3-5(7)4-10/h1-4H
InChI key
VFVHWCKUHAEDMY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2-Chloro-5-nitrobenzaldehyde undergoes condensation reaction with 2-methyl-1-propenylbenzimidazole to yield (E,Z)-2-(2-chloro-5-nitrostyryl)-1-(1-propenyl)benzimidazole. It reacts with 5-aminopyrazoles to yield symmetrical bispyrazolo[3,4-]pyridines.
Application
2-Chloro-5-nitrobenzaldehyde was used in the synthesis of 5-nitro-2-(1H-pyrrol-1-yl)benzaldehyde.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
D E Bacelo et al.
Acta crystallographica. Section C, Crystal structure communications, 53 ( Pt 7), 907-909 (1997-07-15)
The title compound, C18H14ClN3O2, was synthesized by the condensation of 2-chloro-5-nitrobenzaldehyde with 2-methyl-1-propenylbenzimidazole, and the molecule comprises a 2-chloro-5-nitrobenzene and a 1-(Z)-propenylbenzimidazole. The two aromatic moieties are conjugated through the vinyl group. The dihedral angle between the two rings is
Reactions of 5-amino-1, 2-azoles with aromatic and heterocyclic o-chloroaldehydes:[1+ 1] versus [2+ 1] cyclocondensation.
Abramov MA, et al.
Tetrahedron, 57(44), 9123-9129 (2001)
Morita-Baylis-Hillman route to 4H-pyrrolo [1, 2-a][1] benzazepine derivatives.
Park SP, et al.
Tetrahedron, 65(24), 4703-4708 (2009)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service