206555
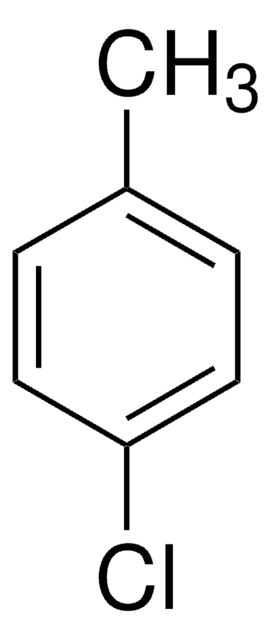
4-Iodotoluene
99%
Synonym(s):
1-Iodo-4-methylbenzene
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
CH3C6H4I
CAS Number:
Molecular Weight:
218.03
Beilstein:
1903637
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
solid
bp
211.5 °C (lit.)
mp
33-35 °C (lit.)
functional group
iodo
SMILES string
Cc1ccc(I)cc1
InChI
1S/C7H7I/c1-6-2-4-7(8)5-3-6/h2-5H,1H3
InChI key
UDHAWRUAECEBHC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
4-Iodotoluene undergoes Suzuki-Miyaura coupling reaction with phenylboronic acid catalyzed by (Ni,Mg)3Si2O5(OH)4 solid-solution nanotubes loaded with palladium. Cobalt-catalyzed coupling of 4-iodotoluene with thiophenols and alkanethiols has been investigated. Palladium/copper-catalyzed Sonogashira cross-coupling reaction of 4-iodotoluene with phenylacetylene has been studied.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
194.0 °F - closed cup
Flash Point(C)
90 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Takashi Mino et al.
The Journal of organic chemistry, 71(25), 9499-9502 (2006-12-02)
Palladium/copper-catalyzed Sonogashira cross-coupling reaction of aryl halides with a variety of terminal alkynes under amine-free conditions in dimethylformamide (DMF) at 80 degrees C gave internal arylated alkynes using PdCl2(MeCN)2 with phosphine-free hydrazone 2a as a ligand and CuI as the
Wancheng Zhu et al.
Inorganic chemistry, 51(11), 6020-6031 (2012-05-16)
(Ni(1-x),Mg(x))(3)Si(2)O(5)(OH)(4) solid-solution nanotubes (NTs) with tunable compositions were hydrothermally synthesized by altering the molar ratio of Mg(2+) to Ni(2+). The as-synthesized NTs were loaded with sub-0.06 wt % palladium (Pd; ∼0.045 wt %) for Suzuki-Miyaura (SM) coupling reactions between iodobenzene
Ying-Chieh Wong et al.
Organic letters, 8(24), 5613-5616 (2006-11-17)
A new cobalt-catalyzed coupling of aryl halides with thiophenols and alkanethiols is reported. A variety of aryl sulfides can be prepared in excellent yields under mild reaction conditions using 1-2 mol % of CoI2(dppe) and Zn. This new cobalt-catalyzed coupling
V Kolaříková et al.
Dalton transactions (Cambridge, England : 2003), 44(45), 19663-19673 (2015-09-17)
Using three different approaches, racemic 1-(perfluoroalkyl)ethylamines were synthesized from perfluoroalkyl iodides or perfluoroalkanoic acids, and further transformed to the corresponding N,N'-disubstituted ethane-1,2-diimines and ethane-1,2-diamines as mixtures of diastereoisomers. Their cyclization afforded imidazolium or dihydroimidazolium salts, which led to silver or
Zengyan Wei et al.
Nature communications, 5, 3870-3870 (2014-05-16)
The shape-controlled synthesis of nanoparticles was established in single-phase solutions by controlling growth directions of crystalline facets on seed nanocrystals kinetically; however, it was difficult to rationally predict and design nanoparticle shapes. Here we introduce a methodology to fabricate nanoparticles
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service