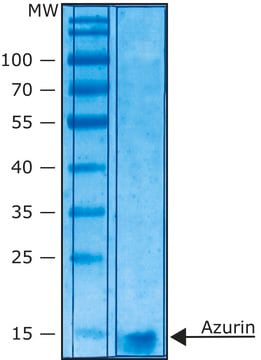
The method of preparation is considered proprietary. However, as mentioned in this properties section of the Product Detail Page, this is Azurin from Pseudomonas aeruginosa. See the links below to review a sample Certificate of Analysis and the product data sheet, respectively:
https://www.sigmaaldrich.com/certificates/sapfs/PROD/sap/certificate_pdfs/COFA/Q14/A3672-1MG0000211689.pdf
https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/306/100/a3672dat.pdf
Select a Size
| Size/SKU | Availability | Price |
|---|
About This Item
biological source
Pseudomonas aeruginosa
Quality Segment
form
lyophilized powder
composition
Protein, ≥65% Lowry
concentration
≥65.0% (Lowry)
technique(s)
toxicology assay: suitable
solubility
water: soluble 1—1.1 mg/mL, clear, blue (light blue to blue)
UniProt accession no.
storage temp.
−20°C
Gene Information
Pseudomonas aeruginosa ... AZU(878046)
General description
Application
- in the cytotoxicity and cell viability studies in human osteosarcoma cell line
- for the functionalization of silicon nitride cantilevers for interaction studies
- for coating gold surface and insulating functionalized oxide surfaces of silicon oxide and mica
Biochem/physiol Actions
Physical form
1 of 1
This Item | |||
|---|---|---|---|
| biological source Pseudomonas aeruginosa | biological source - | biological source insect (firefly) | biological source Pseudomonas aeruginosa (10) |
| technique(s) toxicology assay: suitable | technique(s) toxicology assay: suitable | technique(s) toxicology assay: suitable | technique(s) - |
| concentration ≥65.0% (Lowry) | concentration - | concentration - | concentration - |
| form lyophilized powder | form powder | form solid | form lyophilized powder |
| Gene Information Pseudomonas aeruginosa ... AZU(878046) | Gene Information human ... CCK(885) | Gene Information - | Gene Information - |
| storage temp. −20°C | storage temp. −20°C | storage temp. −20°C | storage temp. 2-8°C |
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
-
How do i know that this is azurin i mean any confirmatory test or anything else.
1 answer-
Helpful?
-