346225
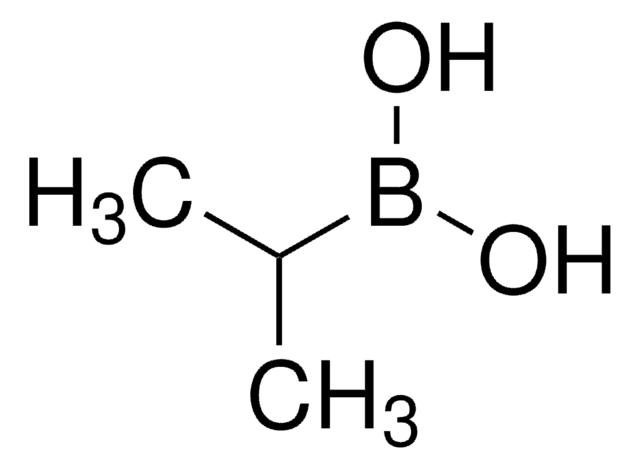
(2-Methylpropyl)boronic acid
≥95.0%
Synonym(s):
Isobutaneboronic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(CH3)2CHCH2B(OH)2
CAS Number:
Molecular Weight:
101.94
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥95.0%
form
solid
mp
108-111 °C (lit.)
SMILES string
CC(C)CB(O)O
InChI
1S/C4H11BO2/c1-4(2)3-5(6)7/h4,6-7H,3H2,1-2H3
InChI key
ZAZPDOYUCVFPOI-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
(2-Methylpropyl)boronic acid can be used as:
It can also be used as a reactant in:
- A reactant in the preparation of 4-isobutylisoquinoline from 4-bromoisoquinoline by Suzuki-Miyaura type couple reaction.
- A catalyst along with aluminum hydroxide, boric acid in the polymerization of styrene.
It can also be used as a reactant in:
- Copper catalyzed cross-coupling reactions.
- The synthesis of polyborylalkanes by Ir-catalyzed C-H borylation reaction.
- The preparation of heterosubstituted diazaboroles and borinines.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Takeshi Yamamoto et al.
Organic letters, 21(16), 6235-6240 (2019-08-07)
Pyrazolylaniline serves as a temporary directing group attached to the boron atom of alkylboronic acids in Ir-catalyzed C(sp3)-H borylation. The reaction takes place at α-, β-, and γ-C-H bonds, giving polyborylated products including di-, tri-, tetra-, and even pentaborylalkanes. α-C-H
Solution-state 15N NMR and solid-state single-crystal XRD study of heterosubstituted diazaboroles and borinines prepared via an effective and simple microwave-assisted solvent-free synthesis
Slabber CA, et al.
Journal of Organometallic Chemistry, 723, 122-128 (2013)
Benjamin M Reeves et al.
Angewandte Chemie (International ed. in English), 58(44), 15697-15701 (2019-09-06)
A transition-metal-free reductive hydroxymethylation reaction has been developed, enabling the preparation of tetrahydroisoquinolines bearing C4-quaternary centers from the corresponding isoquinolines. Deuterium labelling studies and control experiments enable a potential mechanism to be elucidated which features a key Cannizzaro-type reduction followed
Study of Rate-accelerating of Aluminum Hydroxide, Boric Acid, and (2-Methylpropyl) Boronic Acid for Atom Transfer Radical Polymerization of Styrene
Luo Yu-tai, et al.
Journal of Xiamen University (Natural Science), 47(1), 63-63 (2008)
Steven A Rossi et al.
Organic letters, 15(9), 2314-2317 (2013-04-25)
For the first time, a general catalytic procedure for the cross-coupling of primary amides and alkylboronic acids is demonstrated. The key to the success of this reaction was the identification of a mild base (NaOSiMe3) and oxidant (di-tert-butyl peroxide) to
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service