488216
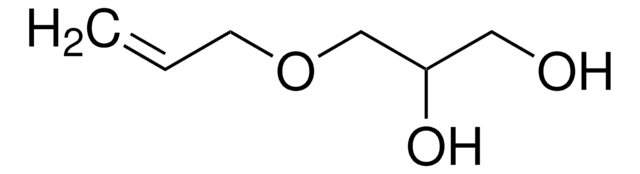
3,4-Dihydroxy-1-butene
≥99%
Synonym(s):
3-Butene-1,2-diol
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
CH2=CHCH(OH)CH2OH
CAS Number:
Molecular Weight:
88.11
Beilstein:
1633578
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥99%
bp
195 °C/733 mmHg (lit.)
density
1.047 g/mL at 25 °C (lit.)
SMILES string
OCC(O)C=C
InChI
1S/C4H8O2/c1-2-4(6)3-5/h2,4-6H,1,3H2
InChI key
ITMIAZBRRZANGB-UHFFFAOYSA-N
Related Categories
General description
3,4-Dihydroxy-1-butene, also known as 3-butene-1,2-diol (BDdiol), is a metabolite of 1,3-butadiene. It forms the precursor for synthesizing different chiral building blocks. BDdiol can undergo oxidation to form hydroxymethylvinyl ketone (HMVK). 1,2-epoxy-3-butene (EB) on hydrolysis in the presence of epoxide hydrolases (EH) forms BDdiol.
Application
3,4-Dihydroxy-1-butene can be used:
- As a reactant to synthesize cyclic organic carbonates by continuous flow procedure.
- To prepare substituted oxazolidinone ligands used to target medicinally relevant RNAs.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Versatile and scalable synthesis of cyclic organic carbonates under organocatalytic continuous flow conditions
Gerardy R, et al.
Catalysis Science & Technology, 9(24), 6841-6851 (2019)
3-Butene-1, 2-diol: An attractive precursor for the synthesis of enantiomerically pure organic compounds.
Rao AVR, et al.
Tetrahedron, 45(22), 7031-7040 (1989)
Christopher L Sprague et al.
Chemical research in toxicology, 17(6), 819-826 (2004-06-23)
3-Butene-1,2-diol (BDD), a major metabolite of 1,3-butadiene (BD), can readily be oxidized to hydroxymethylvinyl ketone (HMVK), a Michael acceptor. In previous studies, 4-(N-acetyl-l-cystein-S-yl)-1,2-dihydroxybutane (DHB), a urinary metabolite of BD that was used to assess human BD exposure, was suggested to
J G Filser et al.
Chemico-biological interactions, 135-136, 249-265 (2001-06-09)
First-pass metabolism of 1,3-butadiene (BD) leading to 1,2-epoxy-3-butene (EB), 1,2:3,4-diepoxybutane (DEB), 3-butene-1,2-diol (B-diol), 3,4-epoxy-1,2-butanediol (EBD) and crotonaldehyde (CA) was studied quantitatively in the once-through BD perfused liver of mouse and rat by means of an all-glass gas-tight perfusion system. Metabolites
Crina M Orac et al.
Journal of medicinal chemistry, 54(19), 6786-6795 (2011-08-05)
The enantiomers and the cis isomers of two previously studied 4,5-disubstituted oxazolidinones have been synthesized, and their binding to the T-box riboswitch antiterminator model RNA has been investigated in detail. Characterization of ligand affinities and binding site localization indicates that
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service