K2144
Ko143 hydrate
≥98% (HPLC), powder, BCRP inhibitor
Synonyme(s) :
(3S,6S,12aS)-1,2,3,4,6,7,12,12a-Octahydro-9-methoxy-6-( 2-methylpropyl)-1,4-dioxopyrazino[1′,2′:1,6]pyrido[3,4- b]indole-3-propanoic acid 1,1-dimethylethyl ester hydrate, Ko-143 hydrate
About This Item
Produits recommandés
product name
Ko143 hydrate, ≥98% (HPLC)
Niveau de qualité
Pureté
≥98% (HPLC)
Forme
powder
Couleur
white to off-white
Solubilité
DMSO: >10 mg/mL
Auteur
GlaxoSmithKline
Température de stockage
room temp
Chaîne SMILES
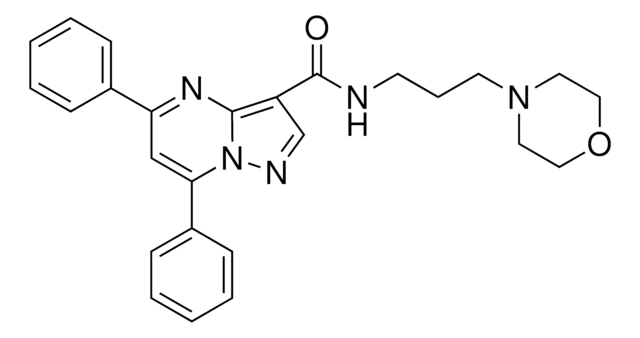
O.COc1ccc2c3C[C@@H]4N([C@@H](CC(C)C)c3[nH]c2c1)C(=O)[C@H](CCC(=O)OC(C)(C)C)NC4=O
InChI
1S/C26H35N3O5.H2O/c1-14(2)11-20-23-17(16-8-7-15(33-6)12-19(16)27-23)13-21-24(31)28-18(25(32)29(20)21)9-10-22(30)34-26(3,4)5;/h7-8,12,14,18,20-21,27H,9-11,13H2,1-6H3,(H,28,31);1H2/t18-,20-,21-;/m0./s1
Clé InChI
ZXXZDXPNJJZRDC-PSLBYKFTSA-N
Application
- to determine the role of ATP-binding cassette sub-family G member 2 (ABCG2), human embryonic kidney (HEK)-C1 and HEK-ABCG2 in tumor microenvironment
- to inhibit ABCG2 for sphere formation assay
- in calcein-AM efflux inhibition to monitor multidrug resistance protein (MRP)-function in kidney
- for cell viability assay
Actions biochimiques/physiologiques
Caractéristiques et avantages
Stockage et stabilité
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Respiratory system
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique






![MK571, Sodium Salt A selective, competitive antagonist of leukotriene D4 (LTD4) (Ki = 2.1 nM for inhibition of [³H]LTD4 binding to human lung membranes).](/deepweb/assets/sigmaaldrich/product/images/110/905/114b43f4-ee35-4aba-ab41-09f068177368/640/114b43f4-ee35-4aba-ab41-09f068177368.jpg)