89339
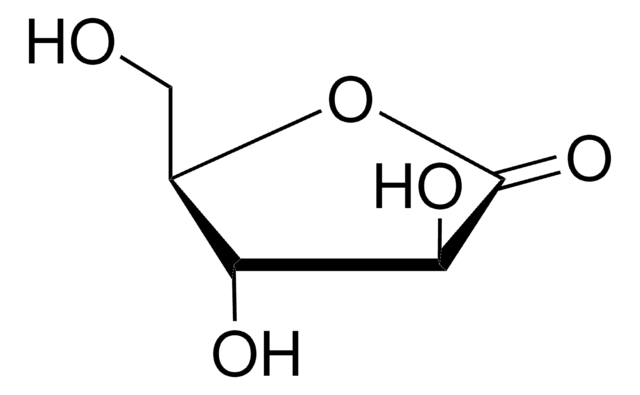
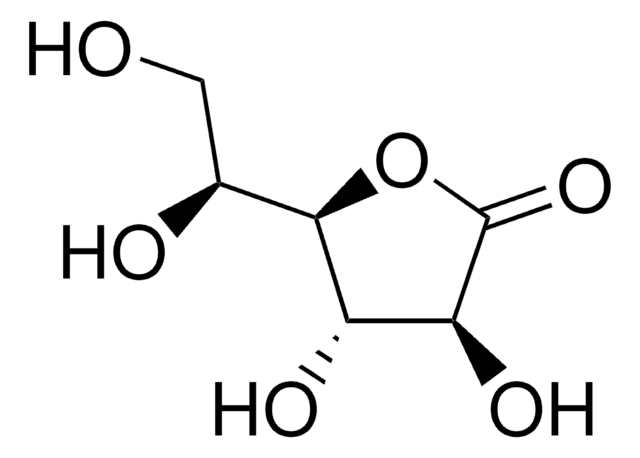
D-Xylono-1,4-lactone
≥95.0% (GC)
Synonym(s):
D-Xylonic acid γ-lactone, D-Xylono γ-lactone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H8O5
CAS Number:
Molecular Weight:
148.11
MDL number:
UNSPSC Code:
12352201
PubChem Substance ID:
NACRES:
NA.25
Recommended Products
Assay
≥95.0% (GC)
form
crystals
suitability
conforms to structure for Proton NMR spectrum
SMILES string
O=C1[C@H](O)[C@@H](O)[C@@H](CO)O1
InChI
1S/C5H8O5/c6-1-2-3(7)4(8)5(9)10-2/h2-4,6-8H,1H2/t2-,3+,4-/m1/s1
InChI key
CUOKHACJLGPRHD-FLRLBIABSA-N
Other Notes
To gain a comprehensive understanding of our extensive range of Monosaccharides for your research, we encourage you to visit our Carbohydrates Category page.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
325.0 °F
Flash Point(C)
162.79 °C
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Structure-based function discovery of an enzyme for the hydrolysis of phosphorylated sugar lactones.
Dao Feng Xiang et al.
Biochemistry, 51(8), 1762-1773 (2012-02-09)
Two enzymes of unknown function from the cog1735 subset of the amidohydrolase superfamily (AHS), LMOf2365_2620 (Lmo2620) from Listeria monocytogenes str. 4b F2365 and Bh0225 from Bacillus halodurans C-125, were cloned, expressed, and purified to homogeneity. The catalytic functions of these
Mervi Toivari et al.
Metabolic engineering, 14(4), 427-436 (2012-06-20)
An NAD(+)-dependent D-xylose dehydrogenase, XylB, from Caulobacter crescentus was expressed in Saccharomyces cerevisiae, resulting in production of 17 ± 2 g D-xylonate l(-1) at 0.23 gl(-1)h(-1) from 23 g D-xylose l(-1) (with glucose and ethanol as co-substrates). D-Xylonate titre and
Harry Boer et al.
AMB Express, 9(1), 48-48 (2019-04-12)
The oxidative D-xylose pathway, i.e. Dahms pathway, can be utilised to produce from cheap biomass raw material useful chemical intermediates. In vitro metabolic pathways offer a fast way to study the rate-limiting steps and find the most suitable enzymes for
Merlin Eric Hobbs et al.
Biochemistry, 52(1), 239-253 (2012-12-12)
A member of the amidohydrolase superfamily, BmulJ_04915 from Burkholderia multivorans, of unknown function was determined to hydrolyze a series of sugar lactones: L-fucono-1,4-lactone, D-arabino-1,4-lactone, L-xylono-1,4-lactone, D-lyxono-1,4-lactone, and L-galactono-1,4-lactone. The highest activity was shown for L-fucono-1,4-lactone with a k(cat) value of
Andreas Reinhardt et al.
Molecular microbiology, 111(4), 1093-1108 (2019-02-02)
The halophilic archaeon Haloferax volcanii utilizes l-rhamnose as a sole carbon and energy source. It is shown that l-rhamnose is taken up by an ABC transporter and is oxidatively degraded to pyruvate and l-lactate via the diketo-hydrolase pathway. The genes
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service