431982
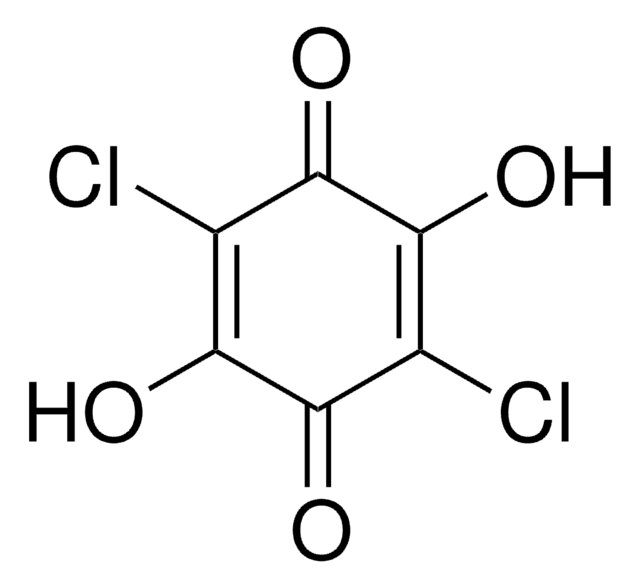
2,6-Dichloro-1,4-benzoquinone
98%
Synonym(s):
2,6-Dichloro-2,5-cyclohexadiene-1,4-dione, 2,6-Dichloro-p-benzoquinone, 2,6-Dichlorobenzoquinone, 2,6-Dichloroquinone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C6H2Cl2O2
CAS Number:
Molecular Weight:
176.98
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
mp
122-124 °C (lit.)
SMILES string
ClC1=CC(=O)C=C(Cl)C1=O
InChI
1S/C6H2Cl2O2/c7-4-1-3(9)2-5(8)6(4)10/h1-2H
InChI key
JCARTGJGWCGSSU-UHFFFAOYSA-N
Related Categories
General description
2,6-Dichloro-1,4-benzoquinone (DCBQ, 2,6-DCBQ) is a halobenzoquinone. Halobenzoquinones are disinfection byproducts (DBPs) found in drinking water. They are capable of producing reactive oxygen species and causing oxidative damage to proteins and DNA in T24 human bladder carcinoma cells. UV-induced transformation of DCBQ in drinking water has been reported. Detection of DCBQ in drinking water by liquid chromatography-ESI tandem mass spectrometry has been reported.
Application
2,6-Dichloro-1,4-benzoquinone is the suitable reagent used to in a study to evaluate the diffusion coefficients (D) for a family of quinones, nitroaromatics, ferrocenes and aromatic hydrocarbon compounds, in acetonitrile by single potential step chronoamperometry. It may be used in the preparation of :
- 2,3,5-Trichloro-1,4-dihydroquinone
- 2,3,5-trichloro-1,4-benzoquinone
- 2,6-Dichloro-1,4-dihydroquinone
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Ben-Zhan Zhu et al.
Proceedings of the National Academy of Sciences of the United States of America, 104(10), 3698-3702 (2007-03-16)
The metal-independent decomposition of organic hydroperoxides and the formation of organic alkoxyl radicals in the absence or presence of halogenated quinones were studied with electron spin resonance (ESR) and the spin-trapping agent 5,5-dimethyl-1-pyrroline N-oxide (DMPO). We found that 2,5-dichloro-1,4-benzoquinone (DCBQ)
Yichao Qian et al.
Environmental science & technology, 47(9), 4426-4433 (2013-04-09)
Halobenzoquinones (HBQs) are a group of emerging disinfection byproducts (DBPs) found in treated drinking water. Because the use of UV treatment for disinfection is becoming more widespread, it is important to understand how the HBQs may be removed or changed
Y I Park et al.
FEBS letters, 444(1), 102-105 (1999-02-26)
Intracellular carbonic anhydrases (CA) in aquatic photosynthetic organisms are involved in the CO2-concentrating mechanism (CCM), which helps to overcome CO2 limitation in the environment. In the green alga Chlamydomonas reinhardtii, this CCM is initiated and maintained by the pH gradient
I Yruela et al.
The Journal of biological chemistry, 266(34), 22847-22850 (1991-12-05)
Oxygen evolution by photosystem II membranes was inhibited by Cu(II) when 2,6-dichlorobenzoquinone or ferricyanide, but not silicomolybdate, was used as electron acceptor. This indicated that Cu(II) affected the reducing side of the photosystem II. The inhibition curves of Cu(II), o-phenanthroline
Denis V Yanykin et al.
Biochimica et biophysica acta, 1797(4), 516-523 (2010-01-26)
Oxygen consumption in Mn-depleted photosystem II (PSII) preparations under continuous and pulsed illumination is investigated. It is shown that removal of manganese from the water-oxidizing complex (WOC) by high pH treatment leads to a 6-fold increase in the rate of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service