推薦產品
化驗
≥98% (HPLC)
形狀
powder
mp
158-162 °C (dec.) (lit.)
溶解度
DMF: 49-51 mg/mL, clear, colorless to faintly yellow
儲存溫度
−20°C
SMILES 字串
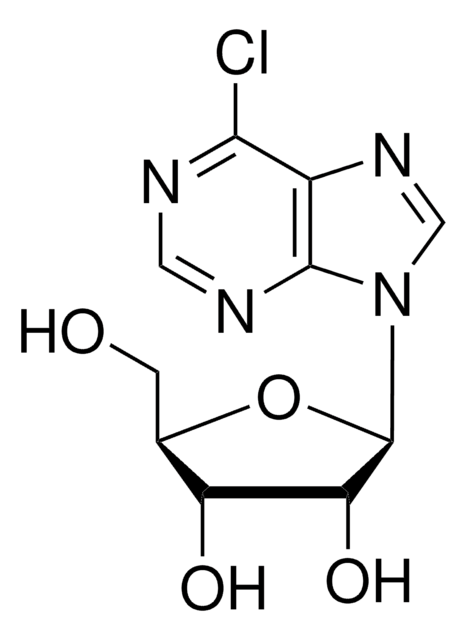
OC[C@H]1O[C@H]([C@H](O)[C@@H]1O)n2cnc3c(Cl)ncnc23
InChI
1S/C10H11ClN4O4/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(18)6(17)4(1-16)19-10/h2-4,6-7,10,16-18H,1H2/t4-,6-,7-,10-/m1/s1
InChI 密鑰
XHRJGHCQQPETRH-KQYNXXCUSA-N
尋找類似的產品? 前往 產品比較指南
應用
6-Chloropurine riboside is used to study the kinetics and substrate specificity of adenosine deaminase. 6-Chloropurine riboside is benzoylated to facilitate synthesis of nucleoside derivatives such as 9-(2,3-Di-deoxy-2-fluoro-β-D-threo-pentofuranosyl)adenine. 6-Chloropurine riboside, especially after phosphorylation to NMP, NDP or NTP, is used as a purine substrate analogue in studies with enzymes such as Inosine monophosphate dehydrogenase (IMPDH); bacteriophage T4 RNA-ligase (EC 6.5.1.3) and pancreatic fibonuclease A.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
L S Singh et al.
Molecular and cellular biochemistry, 204(1-2), 127-134 (2000-03-16)
Adenosine deaminase (ADA) was isolated from small intestine of mice and purified to utmost homogeneity. SDS-PAGE of purified ADA gave a molecular weight of 41 kDa. Western blot analyses gave a single reactive band at 41 kDa and the other
B A Iuodka et al.
Biokhimiia (Moscow, Russia), 58(6), 857-865 (1993-06-01)
The NTP binding site of bacteriophage T4 RNA-ligase (EC 6.5.1.3) was studied using several ATP analogs modified in the purine moiety of the nucleotide at positions 1, 2, 6 and 8: adenosine-N'-oxide 5'-triphosphate (I), 6-chloropurine riboside 5'-triphosphate (II), 6-mercaptopurine riboside
Masahiro Ikejiri et al.
Bioorganic & medicinal chemistry, 15(22), 6882-6892 (2007-09-04)
On the basis of our previous study on antiviral agents against the severe acute respiratory syndrome (SARS) coronavirus, a series of nucleoside analogues whose 5'-hydroxyl groups are masked by various protective groups such as carboxylate, sulfonate, and ether were synthesized
J Alonso et al.
International journal of peptide and protein research, 34(1), 66-69 (1989-07-01)
The reaction of ribonuclease A with either 6-chloropurine riboside 5'-monophosphate or the corresponding nucleoside yields one derivative, with the reagent covalently bound to the alpha-amino group of Lys-1, called derivative II and derivative E, respectively. We studied by means of
R M Richardson et al.
Biochimica et biophysica acta, 953(1), 70-78 (1988-03-02)
When the reaction of bovine pancreatic ribonuclease A with 6-chloropurine riboside 5'-monophosphate was carried out in the presence of several natural mononucleotides, a decrease of 25-75% was found in the amount of the reaction product derivative II (the main product
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務