推薦產品
應用
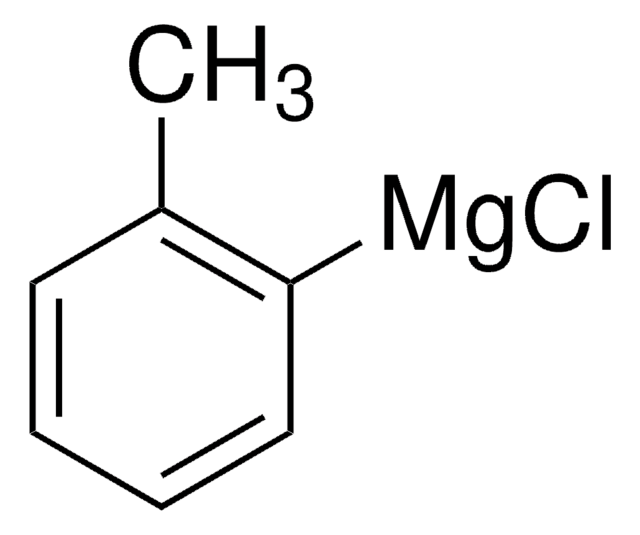
Silicon amine protocol (SLAP) reagents, in conjunction with aldehydes and ketones, form N-unprotected piperazines via photocatalytic cross-coupling with Ir[(ppy)2dtbbpy]PF6 (747769). Heteroaromatic, aromatic, and aliphatic aldehydes are well tolerated, as are structurally and stereochemically complex SLAP reagents. This product was introduced in collaboration with the Bode Research Group and provides a tin-free alternative to SnAP (tin amine protocol) reagents for piperazine synthesis.
其他說明
- Technology Spotlight: SLAP Reagents for Piperazine Synthesis
- Silicon Amine Reagents for the Photocatalytic Synthesis of Piperazines from Aldehydes and Ketones
- Lewis Acid Induced Toggle from Ir(II) to Ir(IV) Pathways in Photocatalytic Reactions: Synthesis of Thiomorpholines and Thiazepanes from Aldehydes and SLAP Reagents.
- Continuous Flow Synthesis of Morpholines and Oxazepanes with Silicon Amine Protocol (SLAP) Reagents and Lewis Acid Facilitated Photoredox Catalysis
相關產品
產品號碼
描述
訂價
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
177.8 °F
閃點(°C)
81 °C
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
Moritz K Jackl et al.
Organic letters, 19(17), 4696-4699 (2017-08-17)
Photocatalytic coupling of aldehydes and silicon amine protocol (SLAP) reagents enables the simple, scalable synthesis of substituted morpholines, oxazepanes, thiomorpholines, and thiazepanes under continuous flow conditions. Key to the success of this process is the combination of an inexpensive organic
Sheng-Ying Hsieh et al.
ACS central science, 3(1), 66-72 (2017-02-06)
Redox neutral photocatalytic transformations often require careful pairing of the substrates and photoredox catalysts in order to achieve a catalytic cycle. This can limit the range of viable transformations, as we recently observed in attempting to extend the scope of
Sheng-Ying Hsieh et al.
Organic letters, 18(9), 2098-2101 (2016-04-22)
Silicon amine protocol (SLAP) reagents for photocatalytic cross-coupling with aldehydes and ketones to form N-unprotected piperazines have been developed. This blue light promoted process tolerates a wide range of heteroaromatic, aromatic, and aliphatic aldehydes and structurally and stereochemically complex SLAP
條款
The expanding class of SLAP reagents provides access to saturated N-heterocycles and aliphatic aldehydes and ketones without additional reagents or the generation of toxic byproducts.
相關內容
The Bode group has developed SnAP (stannyl amine protocol) reagents that cross-couple with aldehydes and ketones to provide one-step access to a wide variety of saturated N-heterocycles.
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務