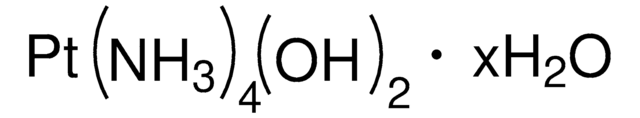SML2811
Phenanthriplatin
>97% (NMR)
Synonym(s):
(SP-4-3)-Diamminechlorido(phenanthridine)platinum(II) nitrate, PhenPt, cis-[Pt(NH3)2(phenanthridine)Cl]NO3
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C13H15ClN4O3Pt
CAS Number:
Molecular Weight:
505.81
UNSPSC Code:
12352200
NACRES:
NA.77
Recommended Products
Quality Level
Assay
>97% (NMR)
form
powder
color
white to beige
solubility
DMSO: 2 mg/mL, clear
storage temp.
2-8°C
SMILES string
Cl[Pt](N)([N+]1=C2C=CC=CC2=C3C=CC=CC3=C1)N.[O-][N+]([O-])=O
Biochem/physiol Actions
Phenanthriplatin is a cationic monofunctional DNA-binding platinum (II) complex with significantly greater anticancer activity (IC50 in μM = 0.035/Ntera2, 0.22/A549, 0.30/HeLa, 0.59/U2OS, 0.74/PC3, 0.94/MCF7, 2.02/HT29; 72 h, MTT assay) than cisplatin, oxaliplatin, and pyriplatin. Enhanced cellular uptake due to its hydrophobic phenanthridine ligand as well as more rapid DNA covalent-binding activity both contribute to its superior anticancer efficacy. Phenanthriplatin binds more effectively to 5′-deoxyguanosine monophosphate than to N-acetyl methionine, whereas pyriplatin reacts equally to both.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Daniele Veclani et al.
Journal of the American Chemical Society, 140(43), 14024-14027 (2018-09-07)
The monofunctional platinum drug phenanthriplatin (phenPt) blocks the replication of cancer cells even if it reacts with only one guanine base. However, there is still insufficient experimental data to improve its cytotoxicity and all previously proposed chemical modifications of the
Jerry D Monroe et al.
PloS one, 13(3), e0192505-e0192505 (2018-03-08)
Unlike cisplatin, which forms bifunctional DNA adducts, monofunctional platinum(II) complexes bind only one strand of DNA and might target cancer without causing auditory side-effects associated with cisplatin treatment. We synthesized the monofunctional triamine-ligated platinum(II) complexes, Pt(diethylenetriamine)Cl, [Pt(dien)Cl]+, and Pt(N,N-diethyldiethylenetriamine)Cl, [Pt(Et2dien)Cl]+
Stuart G Jarrett et al.
Scientific reports, 7(1), 11708-11708 (2017-09-17)
Using primary melanocytes and HEK293 cells, we found that cAMP signaling accelerates repair of bi- and mono-functional platinum-induced DNA damage. Elevating cAMP signaling either by the agonistic MC1R ligand melanocyte stimulating hormone (MSH) or by pharmacologic cAMP induction by forskolin
Wen Zhou et al.
Chemical communications (Cambridge, England), 54(22), 2788-2791 (2018-02-28)
A trans-DDP based monofunctional phenanthridine Pt(ii) complex was synthesized and characterized. Its anticancer activity was studied in vitro on a panel of human cancer cell lines and mouse intestinal cancer organoids. This complex displays significant antitumor properties, with a different
Matthew W Kellinger et al.
Journal of the American Chemical Society, 135(35), 13054-13061 (2013-08-10)
Transcription inhibition by platinum anticancer drugs is an important component of their mechanism of action. Phenanthriplatin, a cisplatin derivative containing phenanthridine in place of one of the chloride ligands, forms highly potent monofunctional adducts on DNA having a structure and
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service