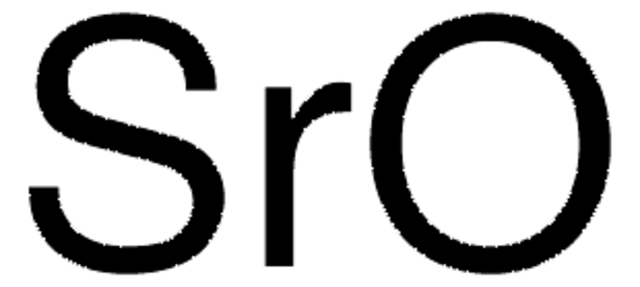472018
Strontium carbonate
≥99.9% trace metals basis
Synonym(s):
Strontium carbonate (SrCO), Strontium monocarbonate 3, Strontium(II) carbonate
About This Item
Recommended Products
Assay
≥99.9% trace metals basis
form
powder
impurities
≤1000.0 ppm Trace Metal Analysis
density
3.7 g/mL at 25 °C (lit.)
SMILES string
[Sr++].[O-]C([O-])=O
InChI
1S/CH2O3.Sr/c2-1(3)4;/h(H2,2,3,4);/q;+2/p-2
InChI key
LEDMRZGFZIAGGB-UHFFFAOYSA-L
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Fabricate ceramic composites for n-type thermoelectrics such as SrTiO3–TiO2.
- Prepare SrCO3/SrO systems for thermochemical energy storage application.
Storage Class Code
13 - Non Combustible Solids
WGK
nwg
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
The prevailing strategies for heat and electric-power production that rely on fossil and fission fuels are having a negative impact on the environment and on our living conditions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service