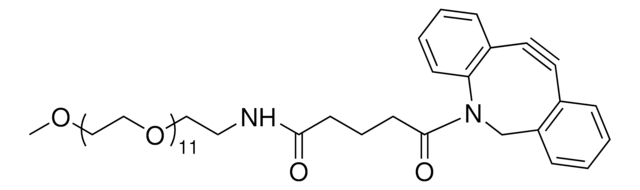
L511188
(S)-2-(2,3-Bis(dicyclohexylamino)cyclopropenimine)-3-phenylpropan-1-ol hydrochloride
AldrichCPR
Synonym(s):
(βS)-β-[[2,3-bis(dicyclohexylamino)-2-cyclopropen-1-ylidene]amino]-benzenepropanol hydrochloride (1:1), Dicyclohexyl cyclopropenimine, Lambert Cyclopropenimine Catalyst
About This Item
Recommended Products
SMILES string
Cl.OC[C@H](Cc1ccccc1)\N=C2\C(N(C3CCCCC3)C4CCCCC4)=C2N(C5CCCCC5)C6CCCCC6
InChI
1S/C36H55N3O.ClH/c40-27-29(26-28-16-6-1-7-17-28)37-34-35(38(30-18-8-2-9-19-30)31-20-10-3-11-21-31)36(34)39(32-22-12-4-13-23-32)33-24-14-5-15-25-33;/h1,6-7,16-17,29-33,40H,2-5,8-15,18-27H2;1H/t29-;/m0./s1
InChI key
RTCSAEOYHXMTHG-JMAPEOGHSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Due to the prevalence of chemical reactions involving proton transfer as a key mechanistic event, Brønsted bases have become indispensable tools for the practice of organic synthetic chemistry, capable of catalyzing proton transfer reactions enantioselectively for the production of optically enriched products.
Novel Brønsted bases provide potent yet tunable basicity to the acidity of a given substrate, are trivial to prepare, and offer unique opportunities for asymmetric transition state organization. The high basicity should allow them to catalyze a wide range of reactions (i.e. chiral pharmaceutical ingredients) and could lead to easier and faster syntheses of novel chiral compounds for drug discovery and other applications.
2,3-bis(dialkylamino)cyclopropenimines serve as a highly effective platform for chiral Brønsted base catalysis. Chiral 2,3-bis(dialkylamino)cyclopropenimine catalyzes the rapid Michael reaction of a glycine imine substrate with high levels of enantioselectivity. Catalysis with chiral cyclopropenimines should be amenable to relatively large-scale applications under mild reaction conditions.

Other Notes
NOTWITHSTANDING ANY CONTRARY PROVISION CONTAINED IN SIGMA-ALDRICH′S STANDARD TERMS AND CONDITIONS OF SALE OR AN AGREEMENT BETWEEN SIGMA-ALDRICH AND BUYER, SIGMA-ALDRICH SELLS THIS PRODUCT "AS-IS" AND MAKES NO REPRESENTATION OR WARRANTY WHATSOEVER WITH RESPECT TO THIS PRODUCT, INCLUDING ANY (A) WARRANTY OF MERCHANTABILITY; (B) WARRANTY OF FITNESS FOR A PARTICULAR PURPOSE; OR (C) WARRANTY AGAINST INFRINGEMENT OF INTELLECTUAL PROPERTY RIGHTS OF A THIRD PARTY; WHETHER ARISING BY LAW, COURSE OF DEALING, COURSE OF PERFORMANCE, USAGE OF TRADE OR OTHERWISE.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service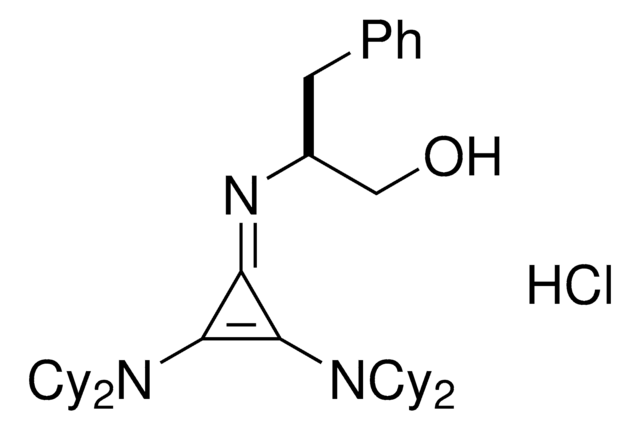


![N-[(1R,2R)-2-(1-Piperidinyl)cyclohexyl]-N′-[4-(trifluoromethyl)phenyl]squaramide 95%](/deepweb/assets/sigmaaldrich/product/structures/238/480/7149c9c0-8769-418a-a96c-77c15dd50cd0/640/7149c9c0-8769-418a-a96c-77c15dd50cd0.png)