E7521
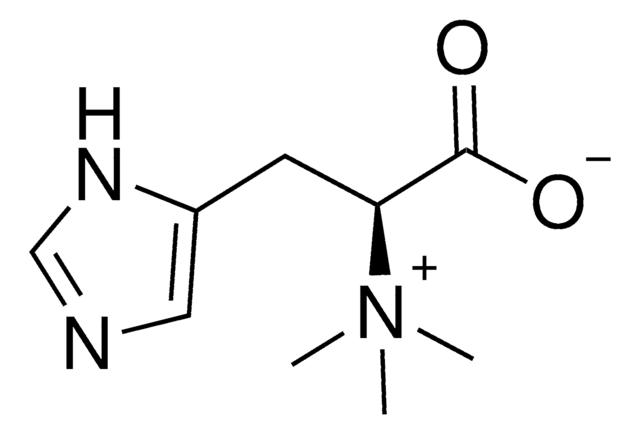
L-(+)-Ergothioneine
Synonym(s):
2-mercaptohistidine trimethyl betaine, Ergothioneine, Sympectothion, Thiasine, Thiolhistidine-betaine, Thioneine, (S)-α-Carboxy-N,N,N-trimethyl-2-mercapto-1H-imidazole-4-ethanaminium inner salt
About This Item
Recommended Products
biological source
fungus (Actinomycetales)
fungus (Ascomycota)
fungus (Basidiomycota)
Assay
≥98.0%
form
powder
mol wt
229.30
storage condition
(Keep container tightly closed in a dry and well-ventilated place)
technique(s)
protein quantification: suitable
solubility
water: 50 mg/mL, clear, colorless
storage temp.
−20°C
SMILES string
C[N+](C)(C)[C@@H](Cc1c[nH]c(S)n1)C([O-])=O
InChI
1S/C9H15N3O2S/c1-12(2,3)7(8(13)14)4-6-5-10-9(15)11-6/h5,7H,4H2,1-3H3,(H2-,10,11,13,14,15)/t7-/m0/s1
InChI key
SSISHJJTAXXQAX-ZETCQYMHSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Research area: Apoptosis
Application
L-(+)-Ergothioneine has been used:
- as a component of the maturation medium for cumulus-oocyte complexes (COCs) to test protective function on lipid peroxide formation
- as an antioxidant compound to test type 2 diabetes patients
- as a positive control in solute carrier protein 22 A4 (SLC22A4) transport assay
Biochem/physiol Actions
Packaging
Other Notes
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service