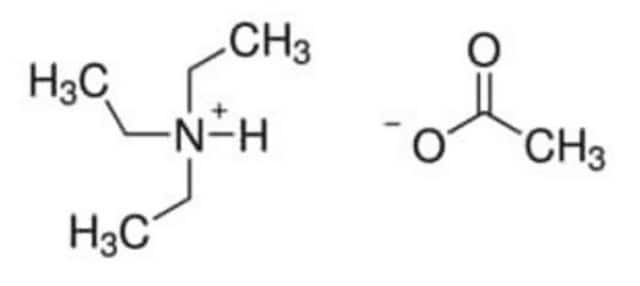
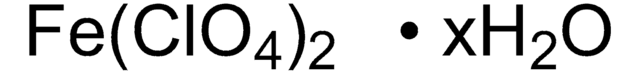420352
Ethylenediamine diacetate
98%
Synonym(s):
Ethanediammonium diacetate
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
NH2CH2CH2NH2 · 2CH3COOH
CAS Number:
Molecular Weight:
180.20
Beilstein:
5444394
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
mp
118-120 °C (lit.)
SMILES string
CC(O)=O.CC(O)=O.NCCN
InChI
1S/C2H8N2.2C2H4O2/c3-1-2-4;2*1-2(3)4/h1-4H2;2*1H3,(H,3,4)
InChI key
SAXQBSJDTDGBHS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Catalytic Synthesis: Utilization of Ethylenediamine Diacetate as an effective catalyst for synthesizing specific thioxothiazolidin-4-one, important for organic chemists and pharmacological studies aiming at new drug development (S Holota, A Lozynskyi, Y Konechnyi, Y Shepeta, 2021).
- Reaction of New Amides: Exploration of reactions between new N-(2,2-Dichloro-1-cyanoethenyl)amides and aliphatic amines facilitated by Ethylenediamine Diacetate, relevant for medicinal chemistry and synthesis (OV Shablykin, SA Chumachenko, 2021).
- Microwave-Assisted Organic Synthesis: Application of Ethylenediamine Diacetate in microwave-assisted synthesis processes, which is valuable for accelerating chemical reactions in drug synthesis and material processing (P Martín-Acosta, G Feresin, A Tapia, 2016).
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Ethylenediamine diacetate (EDDA)-catalyzed one-pot synthesis of tetrahydroquinolines by domino Knoevenagel/hetero Diels-Alder reactions from 1, 3-dicarbonyls.
Lee YR and Hung TV.
Tetrahedron, 67(49), 9627-9634 (2011)
Shima Shahsavari et al.
International journal of radiation biology, 96(4), 502-509 (2019-12-13)
Purpose: Early and accurate imaging of glioblastoma is of great value in staging, metastatic detection, treatment management and prognosis. The human epidermal growth factor receptor 2 (HER2) plays an essential role in the tumorigenesis and tumor progression in cancer. The
Kusum Vats et al.
Journal of labelled compounds & radiopharmaceuticals, 60(9), 431-438 (2017-05-26)
Targeted delivery of chemotherapeutic drug at the tumor site enhances the efficacy with minimum systemic exposure. Towards this, drugs conjugated with peptides having affinity towards a particular molecular target are recognized as affective agents for targeted chemotherapy. Thus, in the
Efficient and general method for the synthesis of benzopyrans by ethylenediamine diacetate-catalyzed reactions of resorcinols with α, β-unsaturated aldehydes. One step synthesis of biologically active (?)-confluentin and (?)-daurichromenic acid.
Lee YR, et al.
Tetrahedron Letters, 46(44), 7539-7543 (2005)
Euan Pyle et al.
Cell chemical biology, 25(7), 840-848 (2018-04-24)
The role of membrane lipids in modulating eukaryotic transporter assembly and function remains unclear. We investigated the effect of membrane lipids in the structure and transport activity of the purine transporter UapA from Aspergillus nidulans. We found that UapA exists
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service