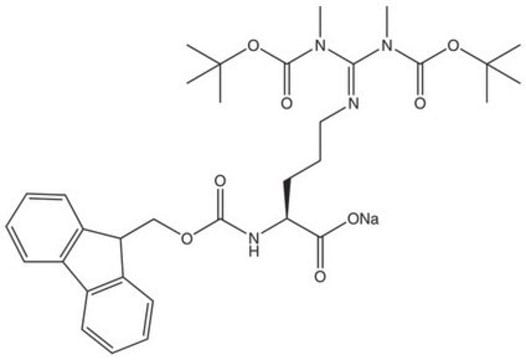
推荐产品
品質等級
產品線
Novabiochem®
化驗
≥95.0% (HPLC)
形狀
powder
反應適用性
reaction type: Fmoc solid-phase peptide synthesis
製造商/商標名
Novabiochem®
應用
peptide synthesis
官能基
Fmoc
儲存溫度
15-25°C
InChI
1S/C35H42N4O7S/c1-20-21(2)31(22(3)27-18-35(4,5)46-30(20)27)47(43,44)39-33(36-6)37-17-11-16-29(32(40)41)38-34(42)45-19-28-25-14-9-7-12-23(25)24-13-8-10-15-26(24)28/h7-10,12-15,28-29H,11,16-19H2,1-6H3,(H,38,42)(H,40,41)(H2,36,37,39)/t29-/m0/s1
InChI 密鑰
JAUPJPAADXVUGQ-LJAQVGFWSA-N
一般說明
Associated Protocols and Technical Articles
Cleavage and Deprotection Protocols for Fmoc SPPS
Literature references
[1] S. Rothbart, et al. (2012) Methods Enzymol., 512, 107.
聯結
分析報告
Appearance of substance (visual): powder
Identity (IR): passes test
Enantiomeric purity: ≥ 99.0 % (a/a)
Purity (DC (018A)): ≥ 95 %
Assay (HPLC, area%): ≥ 95.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
To see the solvent systems used for TLC of Novabiochem® products please click here.
法律資訊
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 2
閃點(°F)
Not applicable
閃點(°C)
Not applicable
商品
Unnatural amino acids, the non-proteinogenic amino acids that either occur naturally or are chemically synthesized, are becoming more and more important as tools for modern drug discovery research.
Unnatural amino acids, the non-proteinogenic amino acids that either occur naturally or are chemically synthesized, are becoming more and more important as tools for modern drug discovery research.
Unnatural amino acids, the non-proteinogenic amino acids that either occur naturally or are chemically synthesized, are becoming more and more important as tools for modern drug discovery research.
Unnatural amino acids, the non-proteinogenic amino acids that either occur naturally or are chemically synthesized, are becoming more and more important as tools for modern drug discovery research.
实验方案
We provide an overview of our available reagents, together with recommendations and details of their use for synthesis of peptides containing post-translationally modified amino acids.
We provide an overview of our available reagents, together with recommendations and details of their use for synthesis of peptides containing post-translationally modified amino acids.
We provide an overview of our available reagents, together with recommendations and details of their use for synthesis of peptides containing post-translationally modified amino acids.
We provide an overview of our available reagents, together with recommendations and details of their use for synthesis of peptides containing post-translationally modified amino acids.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门