795682
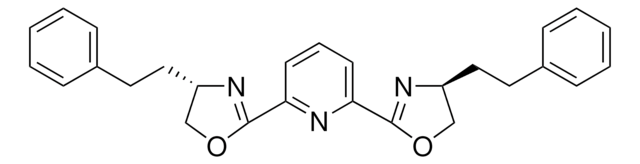
(4R,4′R,5S,5′S)-2,2′-(1-Methylethylidene
95%
Synonym(s):
2,2-Bis[2-(4R,5S-diphenyl-1,3-oxazolinyl)]propane
About This Item
Recommended Products
Quality Level
Assay
95%
form
solid
mp
164-169 °C
SMILES string
CC(C1=N[C@H](C2=CC=CC=C2)[C@H](C3=CC=CC=C3)O1)(C)C4=N[C@H](C5=CC=CC=C5)[C@H](C6=CC=CC=C6)O4
InChI
1S/C33H30N2O2/c1-33(2,31-34-27(23-15-7-3-8-16-23)29(36-31)25-19-11-5-12-20-25)32-35-28(24-17-9-4-10-18-24)30(37-32)26-21-13-6-14-22-26/h3-22,27-30H,1-2H3/t27-,28-,29+,30+/m1/s1
InChI key
ZWWGNCSTEMMQOQ-XAZDILKDSA-N
Application
- To prepare selective exo-catalysts for enantioselective 1,3-dipolar cycloaddition reactions.
- In the asymmetric aminooxygenation of alkenes in the presence of tetramethylaminopyridyl radical (TEMPO) as an oxidant and copper(II) triflate as a catalyst.
- In asymmetric aminofluorination of olefins using an iron catalyst.
This chiral Box ligand was most recently shown to mediate an asymmetric aminofluorination of olefins utlizing Xtalfuor-E (719439) and TREAT-HF (344648). The resulting cyclic carbamates can be readily converted into their concomitant beta-fluoro amino acids.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![2,6-Bis[(3aR,8aS)-(+)-8H-indeno[1,2-d]oxazolin-2-yl)pyridine ≥94%](/deepweb/assets/sigmaaldrich/product/structures/123/619/565288e2-e1c9-4825-a440-17e786bc2c27/640/565288e2-e1c9-4825-a440-17e786bc2c27.png)
![(−)-2,2′-Isopropylidenebis[(4S)-4-phenyl-2-oxazoline] 97%](/deepweb/assets/sigmaaldrich/product/structures/297/720/a29f61c3-34e4-410c-acdd-241699b80af3/640/a29f61c3-34e4-410c-acdd-241699b80af3.png)
![2,2′-Isopropylidenebis[(4S)-4-tert-butyl-2-oxazoline] 99%](/deepweb/assets/sigmaaldrich/product/structures/334/357/19788a81-5365-46fd-978b-6b98382b1117/640/19788a81-5365-46fd-978b-6b98382b1117.png)
![2,6-Bis[(4S)-4-phenyl-2-oxazolinyl]pyridine 98%](/deepweb/assets/sigmaaldrich/product/structures/372/262/fb5c79fe-8277-48b0-a73e-4124c7c2c41c/640/fb5c79fe-8277-48b0-a73e-4124c7c2c41c.png)
![2,2′-Bis[(4S)-4-benzyl-2-oxazoline] 98%](/deepweb/assets/sigmaaldrich/product/structures/139/783/42da3c77-52af-401b-8525-35d96415e284/640/42da3c77-52af-401b-8525-35d96415e284.png)
![(+)-2,2′-Isopropylidenebis[(4R)-4-phenyl-2-oxazoline] 97%](/deepweb/assets/sigmaaldrich/product/structures/232/241/07f8baaa-0ba2-49e0-8ac2-f6d256fb2c84/640/07f8baaa-0ba2-49e0-8ac2-f6d256fb2c84.png)
![[3aR-[2(3′aR*,8′aS*),3′aβ,8′aβ]]-(+)-2,2′-Methylenebis[3a,8a-dihydro-8H-indeno[1,2-d]oxazole] 98%](/deepweb/assets/sigmaaldrich/product/structures/134/031/294d2464-1571-4514-8e4c-c0cda1c1df7b/640/294d2464-1571-4514-8e4c-c0cda1c1df7b.png)
![2,2′-Methylenebis[(4S)-4-phenyl-2-oxazoline] 97%](/deepweb/assets/sigmaaldrich/product/structures/255/350/4403d4f8-c973-4da7-a5b6-2e93d1eacb10/640/4403d4f8-c973-4da7-a5b6-2e93d1eacb10.png)
![2,6-Bis[(4R)-(+)-isopropyl-2-oxazolin-2-yl]pyridine 99%](/deepweb/assets/sigmaaldrich/product/structures/349/609/8673c46e-368a-47a6-a9bd-52bbe13d490a/640/8673c46e-368a-47a6-a9bd-52bbe13d490a.png)

![(4S)-(+)-Phenyl-α-[(4S)-phenyloxazolidin-2-ylidene]-2-oxazoline-2-acetonitrile 97%](/deepweb/assets/sigmaaldrich/product/structures/117/759/d4e6e882-8577-4dcf-84fe-506633ae811a/640/d4e6e882-8577-4dcf-84fe-506633ae811a.png)
![2,2′-Methylenebis[6-(2H-benzotriazol-2-yl)-4-(1,1,3,3-tetramethylbutyl)phenol] 99%](/deepweb/assets/sigmaaldrich/product/structures/236/824/ce89085c-b9e1-4ea0-8157-44b6f9466ed6/640/ce89085c-b9e1-4ea0-8157-44b6f9466ed6.png)