모든 사진(1)
About This Item
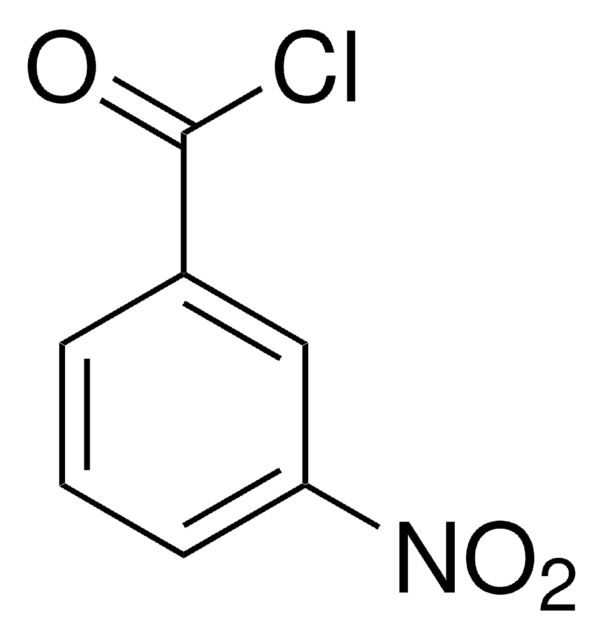
Linear Formula:
CH3C6H3(NO2)COCl
CAS Number:
Molecular Weight:
199.59
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
분석
99%
형태
liquid
refractive index
n20/D 1.581 (lit.)
bp
185 °C/36 mmHg (lit.)
mp
20-21 °C (lit.)
density
1.37 g/mL at 25 °C (lit.)
SMILES string
Cc1ccc(cc1[N+]([O-])=O)C(Cl)=O
InChI
1S/C8H6ClNO3/c1-5-2-3-6(8(9)11)4-7(5)10(12)13/h2-4H,1H3
InChI key
DXMHBBURYDVYAI-UHFFFAOYSA-N
일반 설명
4-Methyl-3-nitrobenzoyl chloride is a benzoyl chloride derivative. It has been reported to be synthesized from 4-methyl-3-nitrobenzoic acid.
애플리케이션
4-Methyl-3-nitrobenzoyl chloride was used in the synthesis of 4-amino-1,5-naphthalenedisulphonate acid monosodium salt, an intermediate employed in the synthesis of modified suramin molecule.
It may be used in the synthesis of the following:
It may be used in the synthesis of the following:
- benzophenone derivative
- substituted 3-amino-4-methyl-N-phenylbenzamide
- retroamide
- 4-methyl-3-nitro-N-phenylbenzamide
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point (°F)
235.4 °F - closed cup
Flash Point (°C)
113 °C - closed cup
개인 보호 장비
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
M J Ashton et al.
Journal of medicinal chemistry, 39(17), 3343-3356 (1996-08-16)
The synthesis and biological activity of a new series of benzamides and related compounds that upregulate the expression of the low-density lipoprotein (LDL) receptor in human hepatocytes (HepG2 cells) by a novel mechanism are described. The lead compound, N-[5-[(3-cyclohexylpropionyl)amino]-2-methylphenyl]-4-hydroxybe nzamide
Holly L Deak et al.
Bioorganic & medicinal chemistry letters, 18(3), 1172-1176 (2007-12-18)
N-3-(Phenylcarbamoyl)arylpyrimidine-5-carboxamides are a novel class of selective Lck inhibitors. This series of compounds derives its selectivity from a hydrogen bond with the gatekeeper Thr316 of the enzyme. X-ray co-crystal structural data, structure-activity relationships, and the synthesis of these inhibitors are
Shuxin Li et al.
Bioorganic & medicinal chemistry letters, 22(16), 5279-5282 (2012-07-14)
A series of acrylamide analogues were designed and synthesized from Imatinib and Nilotinib as novel BCR-ABL inhibitors by application of the principle of nonclassical electronic isostere. All new compounds were evaluated for their inhibitory effects on the activity of BCR-ABL
Romina Croci et al.
PloS one, 9(3), e91765-e91765 (2014-03-14)
Noroviruses (NV) are +ssRNA viruses responsible for severe gastroenteritis; no effective vaccines/antivirals are currently available. We previously identified Suramin (9) as a potent inhibitor of NV-RNA dependent RNA polymerase (NV-RdRp). Despite significant in vitro activities versus several pharmacological targets, Suramin
Marie-Pierre Lézé et al.
Bioorganic & medicinal chemistry letters, 18(16), 4713-4715 (2008-07-22)
Two new series of benzonitrile derivatives on position 6 or 4 of indole ring were successfully synthesized via a Leimgruber-Batcho reaction. All the compounds were evaluated in vitro on the inhibition of aromatase (CYP19) and 17alpha-hydroxylase-C17,20-lyase (CYP17). The racemate, 4-[(1H-imidazol-1-yl)(1H-indol-4-yl)methyl]benzonitrile
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.