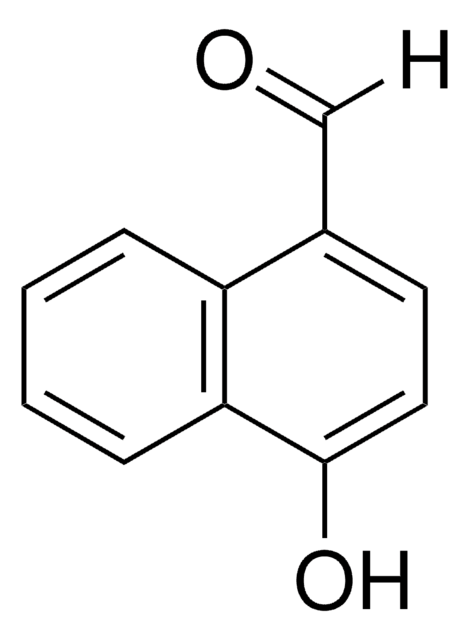
推荐产品
化驗
97%
mp
32-36 °C (lit.)
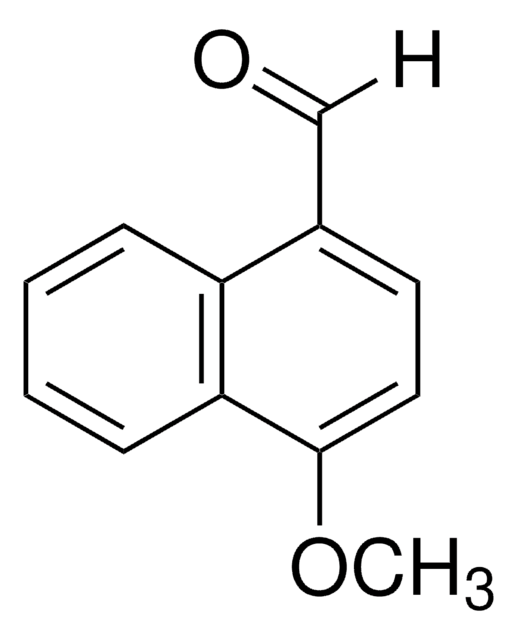
SMILES 字串
Cc1ccc(C=O)c2ccccc12
InChI
1S/C12H10O/c1-9-6-7-10(8-13)12-5-3-2-4-11(9)12/h2-8H,1H3
InChI 密鑰
LANRGTXVAPBSIA-UHFFFAOYSA-N
一般說明
4-Methyl-1-naphthaldehyde is a monoaldehyde. It is obtained along with 1-methyl-2-naphthaldehyde from 1-methylnaphthalene, via formylation.
應用
4-Methyl-1-naphthaldehyde may be used in the synthesis of methyl 2-phenyl-3-( 4-methyl-1-naphthyl)propenoate and (Z/E)-2-amino-4,6-dimethyl-5-[(4-methyl-1-naphthyl)methylene]-5H-cyclopenta[b]pyridine-3,7-dicarbonitrile.
儲存類別代碼
13 - Non Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
230.0 °F - closed cup
閃點(°C)
110 °C - closed cup
個人防護裝備
Eyeshields, Gloves, type N95 (US)
S Amin et al.
Chemical research in toxicology, 1(6), 349-355 (1988-11-01)
Previous studies have shown that 5-methylchrysene (5-MeC) is more carcinogenic on mouse skin than the other methylchrysenes and that the structural requirements favoring tumorigenicity of methylated polynuclear aromatic hydrocarbons are the presence of a bay region methyl group and free
The influence of aromatic compound protonation on the regioselectivity of Gattermann-Koch formylation.
Tanaka M, et al.
Chemical Communications (Cambridge, England), 2, 159-160 (1996)
Stéphanie Kolb et al.
European journal of medicinal chemistry, 45(3), 896-901 (2009-12-09)
We report herein the synthesis of 5-substituted [1]pyrindine derivatives and the evaluation of their antiproliferative properties on HeLa cells, a cervical carcinoma tumor cell line, and on the melanoma A2058 cell line. The most efficient compounds display cytotoxicity against tumor
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门