901494
N-Methylated pomalidomide
≥98%
Sinónimos:
4-Amino-2-(1-methyl-2,6-dioxopiperidin-3-yl)isoindoline-1,3-dione, E3 Ligase ligand methylated negative control, Ligand for PROTAC® research
About This Item
Productos recomendados
ligand
N-Methylated Pomalidomide
Quality Level
assay
≥98%
form
powder or crystals
reaction suitability
reagent type: ligand
shipped in
wet ice
storage temp.
2-8°C
SMILES string
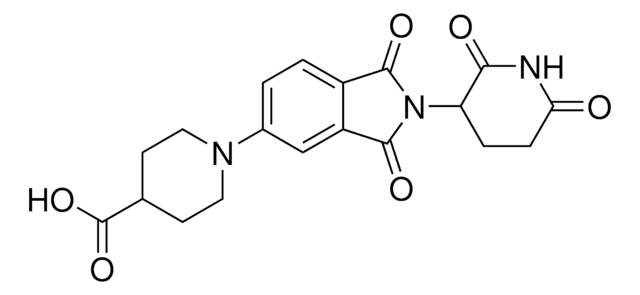
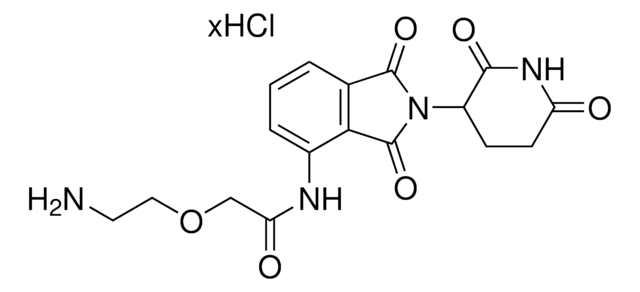
O=C(C(CC1)N(C2=O)C(C3=C2C=CC=C3N)=O)N(C)C1=O
Application
Other Notes
Portal: Building PROTAC® Degraders for Targeted Protein Degradation
Hijacking the E3 Ubiquitin Ligase Cereblon to Efficiently Target BRD4
Targeted Protein Degradation by Small Molecules
Small-Molecule PROTACS: New Approaches to Protein Degradation
Legal Information
related product
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Protein Degrader Building Blocks are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
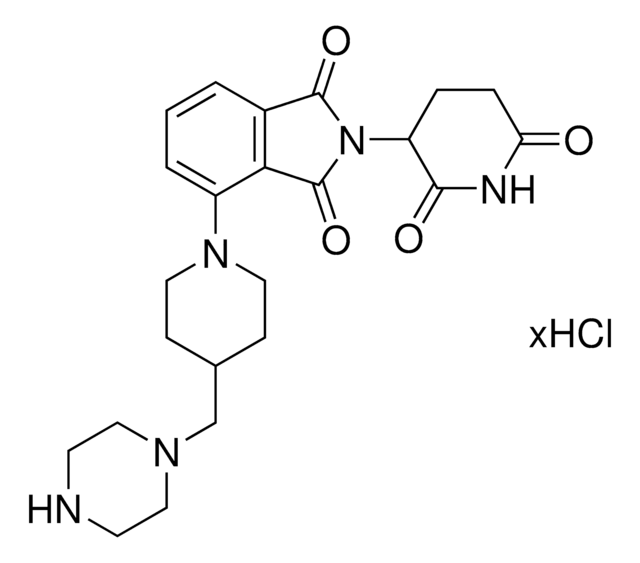
Protein Degrader Building Blocks are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
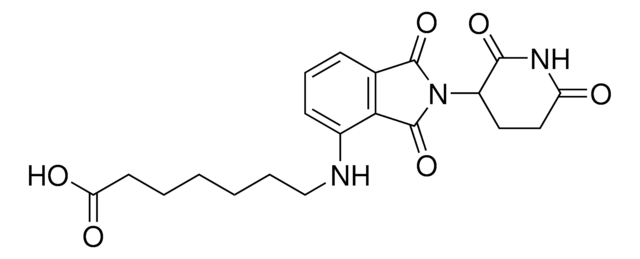
Protein Degrader Building Blocks are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
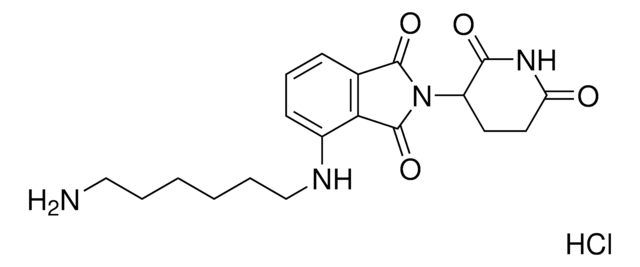
Protein Degrader Building Blocks are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico