903132
1-(Fluorosulfonyl)-2,3-dimethyl-1H-imidazol-3-ium trifluoromethanesulfonate
≥95%
Synonym(s):
1-(Fluorosulfuryl)-2,3-dimethyl-1H-imidazol-3-ium trifluoromethanesulfonate, Fluorosulfuryl imidazolium triflate salt, SuFEx-IT
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C6H8F4N2O5S2
CAS Number:
Molecular Weight:
328.26
UNSPSC Code:
12352101
NACRES:
NA.22
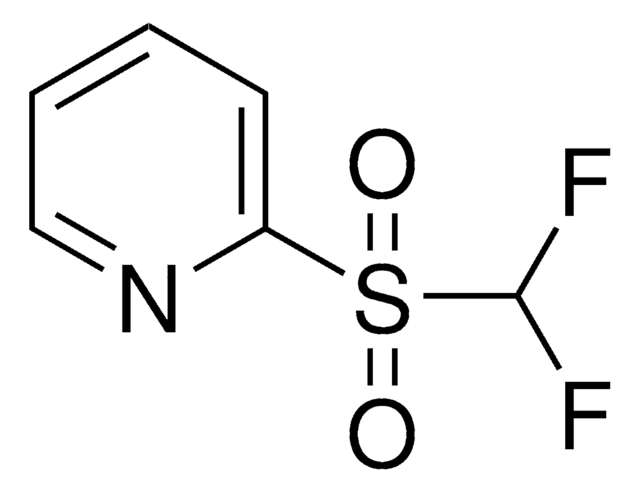
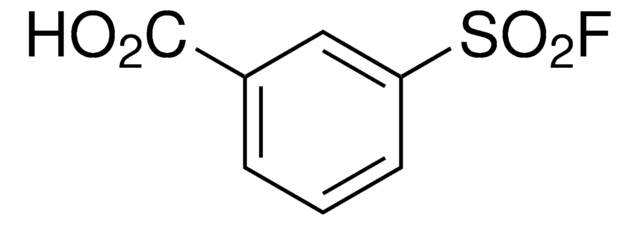
Recommended Products
Assay
≥95%
form
solid
mp
64-65 °C
storage temp.
2-8°C
Application
This solid fluorosulfuryl imidazolium triflate salt, SuFEx-IT, is a convenient alternative to sulfuryl fluoride (SO2F2), which is a harmful fumigant gas with difficult handling procedures that make SO2F2 -- and the products it is capable of making -- difficult to access. However, this reagent possesses novel reactivity, selectivity, and scope and was demonstrated by the labs of K. Barry Sharpless and Jiajia Dong to convert phenols, primary amines, and secondary amines to fluorosulfates and sulfamoyl fluorides. Fluorosulfates are used widely in biological studies, medicinal chemistry, and work as a pseudohalide in Pd-catalysed cross-coupling reactions.
Store in dry environment (desiccator advised) at 4 °C.
Automate your fluorination reactions with Synple Automated Synthesis Platform (SYNPLE-SC002)
Store in dry environment (desiccator advised) at 4 °C.
Automate your fluorination reactions with Synple Automated Synthesis Platform (SYNPLE-SC002)
related product
Product No.
Description
Pricing
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Taijie Guo et al.
Angewandte Chemie (International ed. in English), 57(10), 2605-2610 (2017-12-26)
Sulfuryl fluoride, SO2 F2 , has been found to derivatize phenols in all kinds of environments, even those in highly functional molecules. We now report that a solid fluorosulfuryl imidazolium triflate salt delivers the same "F-SO2 +" fragment to Nu-H
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![4-(Acetylamino)phenyl]imidodisulfuryl difluoride ≥98%](/deepweb/assets/sigmaaldrich/product/structures/101/806/3f40354f-e903-4ea0-9654-10872377816c/640/3f40354f-e903-4ea0-9654-10872377816c.png)



![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)