Biosimilar Comparability Workflow for Middle-Up Mass Analysis Using Trastuzumab as Model Protein
Geoffrey Rule1, Sundaram Palaniswamy2, Uma Sreenivasan3, Paresh Tank4, Fatima D’Souza4
1Bellefonte, PA, USA, 2Bangalore, India, 3Round Rock, TX, USA, 4ZRAS, Zelle Biotechnology Pvt. Ltd., Thane, India
Introduction
Biosimilar mAb drugs have surged in the last decade due to patent expiration of innovator drugs, offering affordable care to patients. Biosimilars, while sharing amino acid sequences with innovators, can differ structurally due to post-translational modifications (PTMs) resulting from changes in manufacturing processes. These variations, like N-glycosylation and alterations to termini, pose challenges in proving comparability.1
Thorough characterization of therapeutic mAbs is vital for safety and efficacy and establishing critical quality attributes (CQAs) are essential for both innovator and biosimilar drugs.2, 3 Characterization generally involves chromatography (e.g., SEC, RP, HILIC) combined with mass spectrometry (MS).
Trastuzumab received approval for medical usage in the United States in September 1998 and in the European Union in August 2000. At present, five biosimilars have gained approval in both the European Union and the United States.4, 5
This note describes the application of a reducing RPLC-MS workflow for middle-up comparison of trastuzumab innovator and biosimilar. The application note also includes the correlation of de-charged masses of respective heavy and light chains with theoretical masses.
A recombinant human antibody standard, SILuTMLite SigmaMAb (MSQC4), is utilized as a reference and assay control sample. Here, this is referred to simply as SigmaMAb although several other SigmaMAb standards are also available commercially.
Workflow for Middle-Up Mass Analysis of Trastuzumab
Antibody reduction (optional)
Chromatography
Measurement and Analysis
An RPLC-MS workflow has been developed to simplify mass analysis of reduced monoclonal antibodies (mAbs) for biosimilarity assessment.
In detail, it includes:
- Antibody reduction procedures
- Mass spectrometer calibration
- System suitability test utilizing a recombinant human monoclonal antibody reference
- An RPLC-MS method compatible with sample separation and analysis of reduced monoclonal antibodies
General Procedures - Sample and Reference Preparation and System Setup
Antibody Reduction Procedure
All mAb samples were solubilized in 100 μL water at a concentration of 1 mg/mL.
Full reduction of samples is performed by this protocol:
- Prepare a 100 mM solution of tris(2-carboxyethyl)phosphine (TCEP) in 6 M aqueous guanidine hydrochloride by dissolving 2.87 g of TCEP and 57.32 g of guanidine hydrochloride in 100 mL of water (if less solution is needed, scale down accordingly).
- Combine 30 μL of the resulting solution with 10 μL of sample.
- Incubate for two hours at 37 °C.
Note: Full reduction is performed in denaturing conditions, where both the inter-chain and intra-chain disulfide bonds are broken. For a partial reduction procedure see the application note Protocol for Purification, Optional Reduction, and SEC-MS Analysis of a Monoclonal Antibody.6
Instrument Calibration
The Waters QToF Xevo G2XS mass spectrometer was calibrated in a mass range of 400 – 4000 m/z with a 10 μL/min infusion of 0.4 mg/mL of sodium iodide in water. Calibration can also be performed with a 10 μL/min infusion of 0.4 mg/mL cesium iodide and polyalanine in water, prior to running the samples.
System Suitability
Reduced SigmaMAb antibody reference (formulated at 2 mg/mL and further diluted to 1 mg/mL) was analyzed alongside samples to determine system suitability.
RPLC-MS System Setup and MS Data Analysis
RPLC-MS System Setup
Essential settings of the UHPLC-PDA chromatography system and qToF mass spectrometer are listed in Tables 1 and 2 below.
MS Data Analysis
Data were processed using the MaxEnt1 module within the UNIFI software to generate deconvoluted mass spectra. In general, a summed spectrum was created from the total ion chromatogram (TIC) of the heavy chain (HC), or light chain (LC), subunits and the spectrum then processed by MaxEnt1; detailed parameters in Table 3. Glycoforms were matched by the software and HC glycans are listed in the Results section. Relative abundance data were tabulated based on peak intensities of the coeluting glycoform species.
Results
Intact Mass Analysis of Reduced Trastuzumab
The objective of this analysis was to compare the molecular weight of trastuzumab innovator and biosimilar, light, and heavy chains, using an RPLC-MS, middle-up approach.
System Suitability Test Results
Four microliters of reduced SigmaMAbTM reference sample was injected onto the RPLC-MS system. The observed heavy chain glycoforms matched the expected glycoform masses of SigmaMAbTM, as listed in Table 4. Discrepancies between the observed masses and the theoretical values for four glycoforms are all within 0.003% mass error. Figure 1 illustrates the UV (280 nm) and TIC chromatograms of the SigmaMAbTM reference, while Figure 2 shows deconvoluted mass spectra of the light and heavy chain glycoforms.
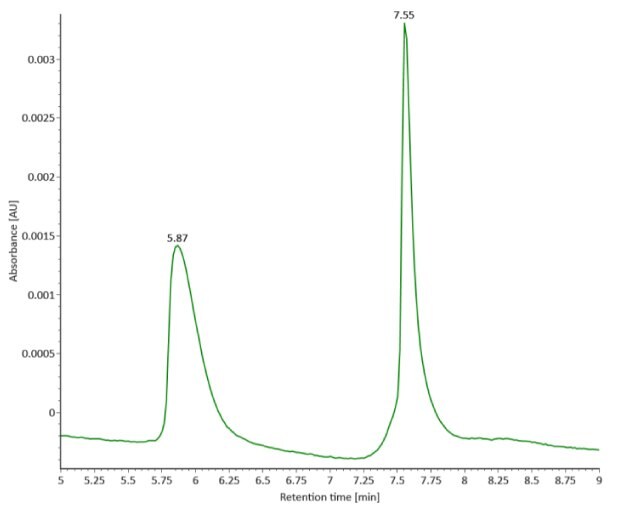
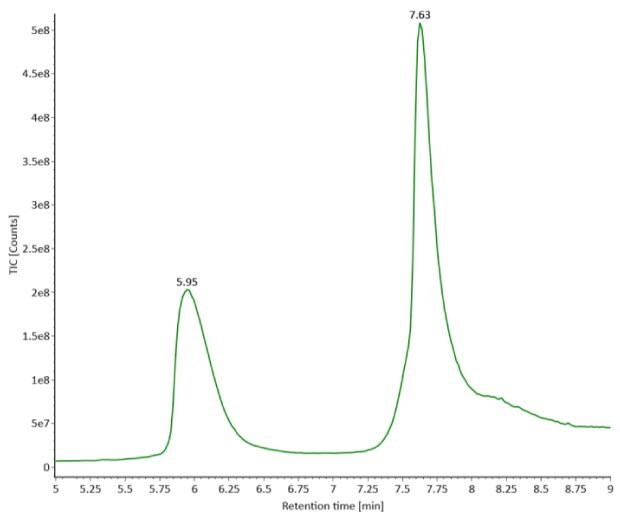
Figure 1. Photodiode array (280 nm, left) and TIC trace (right) of reduced SigmaMAbTM reference.
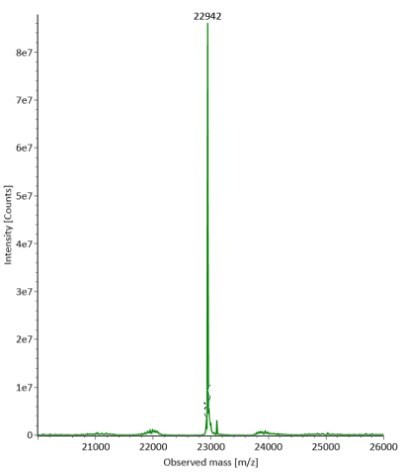
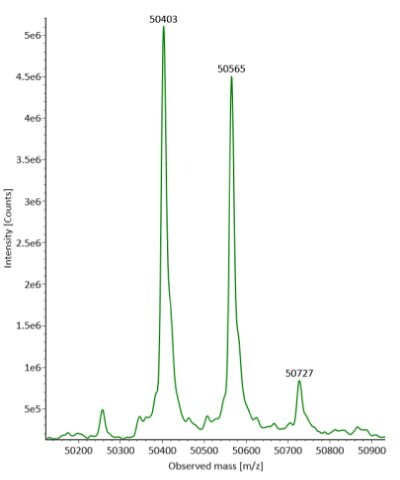
Figure 2. Deconvoluted mass spectra of light and heavy chains (left and right, respectively) of fully reduced SigmaMAbTM reference.
Reduced Sample Results
The fully reduced trastuzumab innovator and biosimilar samples were also analyzed using RPLC-MS. The corresponding photodiode array (280 nm) traces, TICs, and the summed and deconvoluted MS spectra of the samples are shown in Figures 3 and 4. The measured masses of reduced forms correlated well with the calculated masses as shown in Table 5 (mass error 0.001%, or less, for innovator and 0.003%, or less, for biosimilar).


Figure 3. Comparative photodiode array (280 nm, left) and TIC trace (right) of fully reduced trastuzumab. Red: Innovator; Blue: Biosimilar.


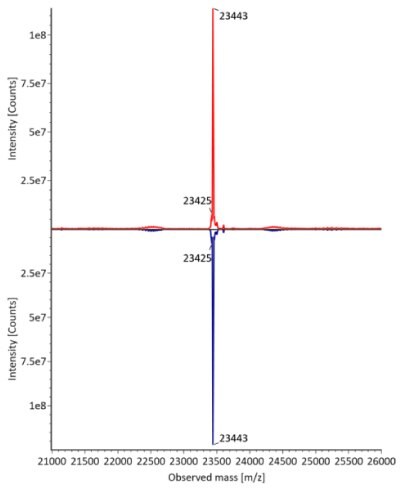
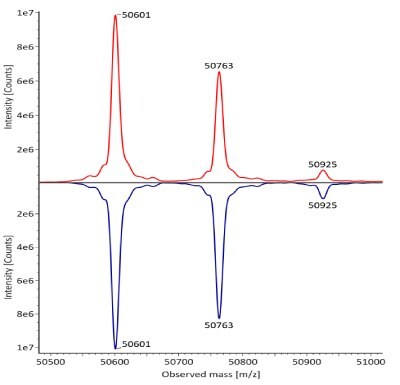
Figure 4. Comparative summed (top) and deconvoluted (bottom) mass spectra of light and heavy chains (left and right, respectively) of fully reduced trastuzumab. Red: Innovator; Blue: Biosimilar.
Conclusion
Ensuring regulatory approvals of biosimilar mAb drug products requires meticulous comparability assessment to guarantee safety and efficacy. This application note utilizes reversed-phase liquid chromatography-mass spectrometry (RPLC-MS) at the subunit level for assessing comparability between originator trastuzumab and its biosimilar counterpart. An RP method suitable for reduced mAbs was established using a wide-pore, superficially porous C4 column.
The workflow uses a mAb reduction procedure, performed with full reduction so that all disulfide bonds are broken. Solutions of sodium iodide, or cesium iodide, were used for mass spectrometer calibration.
Upon analyzing the reduced SigmaMAbTM reference sample, observed glycoform masses of the heavy chain were consistent with anticipated masses with the LC and three HC glycoforms all within a 0.003% mass error threshold. Similarly, the measured masses of reduced innovator trastuzumab, and biosimilar, exhibited close alignment with theoretical masses (mass error of 0.003%, or below).
These findings affirm the versatility of the RPLC-MS workflow for analyzing reduced monoclonal antibody samples, delivering accurate results for distinct identification of multiple glycoforms.
See more applications on the Biopharmaceutical Characterization page.
References
Pour continuer à lire, veuillez vous connecter à votre compte ou en créer un.
Vous n'avez pas de compte ?