All Photos(3)
About This Item
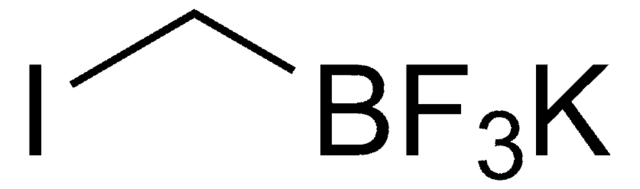
Linear Formula:
BrCH2BF3K
CAS Number:
Molecular Weight:
200.84
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
90%
form
solid
mp
225-230 °C
SMILES string
[K+].F[B-](F)(F)CBr
InChI
1S/CH2BBrF3.K/c3-1-2(4,5)6;/h1H2;/q-1;+1
InChI key
AZDFPIRYUOCVCJ-UHFFFAOYSA-N
Application
Organotrifluoroborate involved in:
Organotrifluoroborates as versatile and stable boronic acid surrogates.
- Suzuki-Miyaura cross-coupling reactions
- Synthesis of functionalized ethyltrifluoroborates
- SN2 displacement with alkoxides
Organotrifluoroborates as versatile and stable boronic acid surrogates.
Versatile starting material for preparation of a variety of functionalized substrates for Suzuki coupling.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Gary A Molander et al.
Organic letters, 8(13), 2767-2770 (2006-06-16)
[reaction: see text] We have successfully prepared potassium azidoalkyltrifluoroborates from the corresponding halogen compounds in 94-98% yields through a nucleophilic substitution reaction with NaN(3). In the presence of various alkynes and Cu(I) as a catalyst, these azidotrifluoroborates easily formed 1,4-disubstituted
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service