All Photos(1)
About This Item
Empirical Formula (Hill Notation):
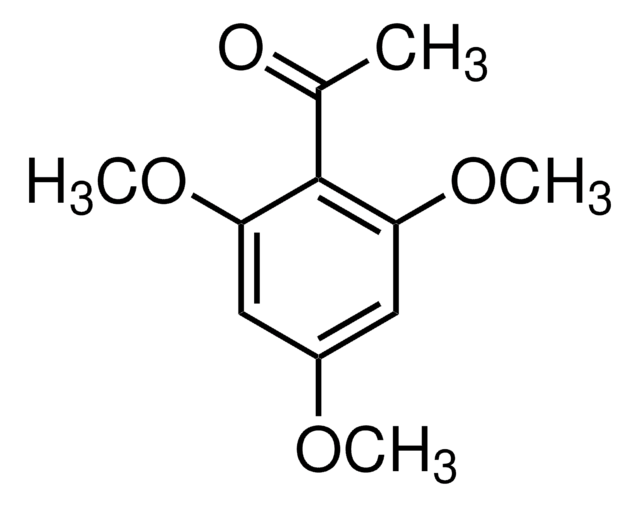
C10H12O4
CAS Number:
Molecular Weight:
196.20
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
solid
mp
80-84 °C (lit.)
SMILES string
COc1cc(O)c(C(C)=O)c(OC)c1
InChI
1S/C10H12O4/c1-6(11)10-8(12)4-7(13-2)5-9(10)14-3/h4-5,12H,1-3H3
InChI key
FBUBVLUPUDBFME-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Rodrigo dos Santos et al.
Archiv der Pharmazie, 339(5), 227-237 (2006-03-31)
Semi-empirical molecular orbital calculations at AM1 level were done with the aim to investigate the structure-activity relationships of antispasmodic activities of ten 2-(X-benzyloxy)-4,6-dimethoxyacetophenones with X = H, 4'-F, 4'-NO2, 4'-CH3, 4'-Cl, 3',4'-(CH3)2, 4'-OCH3, 4'-Br, 4'-OCH2C6H5, and 4'-C(CH3)3, against acetylcholine-induced contraction
Fungicide and fungiostatic effects of xanthoxyline.
V Cechinel Filho et al.
Journal of ethnopharmacology, 53(3), 171-173 (1996-09-01)
C Tringali et al.
Fitoterapia, 72(5), 538-543 (2001-06-29)
Analysis of the polar fractions of an EtOH extract obtained from the bark of the African medicinal plant Fagara macrophylla led to the isolation and identification of the alkaloids oblongine (6), tembetarine (7) and magnoflorine (8) and the flavonoid hesperidin
V Cechinel Filho et al.
Journal of pharmaceutical sciences, 84(4), 473-475 (1995-04-01)
The antispasmodic activity of several xanthoxyline derivatives against acetylcholine-induced contraction of the guinea pig ileum was evaluated in vitro. The acetophenones with two methoxyl groups, mainly in the 3,4 positions, exhibited potent antispasmodic activity. Modification of the hydroxyl group in
J B Calixto et al.
Planta medica, 56(1), 31-35 (1990-02-01)
The inhibitory action of the major constituent of Sebastiania schottiana (Euphorbiaceae), 2-hydroxy-4,6-dimethoxyacetophenone (xanthoxyline) on contractions induced by agonists and electrical stimulation of smooth and cardiac muscle preparations was analysed. Xanthoxyline (30 to 300 microM) inhibited contractions of the rat uterus
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service