PF140
MMP-9, Active, Human, Recombinant
Sinónimos:
Gelatinase B (67 kDa), Matrix Metalloproteinase-9 (67 kDa)
About This Item
Productos recomendados
form
liquid
Quality Level
specific activity
20.0-60.0 ΔA405/h-μg protein (thiopeptide hydrolysis assay)
does not contain
preservative
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
avoid repeated freeze/thaw cycles
shipped in
wet ice
storage temp.
−70°C
General description
Matrix metalloproteinases are members of a unique family of proteolytic enzymes that have a zinc ion at their active sites and can degrade collagens, elastin and other components of the extracellular matrix (ECM). These enzymes are present in normal healthy individuals and have been shown to have an important role in processes such as wound healing, pregnancy, and bone resorption. However, overexpression and activation of MMPs have been linked with a range of pathological processes and disease states involved in the breakdown and remodeling of the ECM. Such diseases include tumor invasion and metastasis, rheumatoid arthritis, periodontal disease, and vascular processes such as angiogenesis, intimal hyperplasia, atherosclerosis and aneurysms. Recently, MMPs have been linked to neurodegenerative diseases such as Alzheimer’s, and amyotrophic lateral sclerosis (ALS). Natural inhibitors of MMPs, tissue inhibitor of matrix metalloproteinases (TIMPs) exist and synthetic inhibitors have been developed which offer hope of new treatment options for these diseases. Regulation of MMP activity can occur at the level of gene expression, including transcription and translation, level of activation, or at the level of inhibition by TIMPs. Thus, perturbations at any of these points can theoretically lead to alterations in ECM turnover. Expression is under tight control by pro- and anti-inflammatory cytokines and/or growth factors and, once produced the enzymes are usually secreted as inactive zymograms. Upon activation (removal of the inhibitory propeptide region of the molecules) MMPs are subject to control by locally produced TIMPs. All MMPs can be activated in vitro with organomercurial compounds (e.g., 4-aminophenylmercuric acetate), but the agents responsible for the physiological activation of all MMPs have not been clearly defined. Numerous studies indicate that members of the MMP family have the ability to activate one another. The activation of the MMPs in vivo is likely to be a critical step in terms of their biological behavior, because it is this activation that will tip the balance in favor of ECM degradation. The hallmark of diseases involving MMPs appear to be stoichiometric imbalance between active MMPs and TIMPs, leading to excessive tissue disruption and often degradation. Determination of the mechanisms that control this imbalance may open up some important therapeutic options of specific enzyme inhibitors.
Application
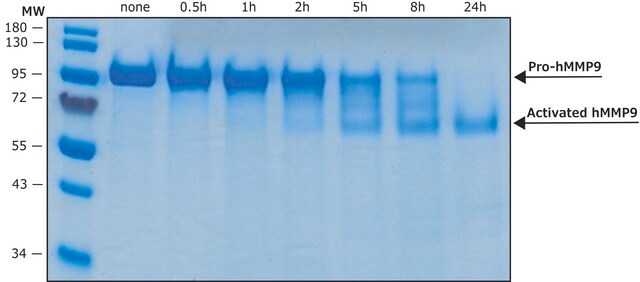
Zymography (0.1 μg protein/lane, see application references)
Packaging
Warning
Physical form
Reconstitution
Other Notes
Backstrom, J.R., et al. 1996. J. Neuro.16, 7910.
Lim, G.P., et al. 1996. J Neurochem.67.
Xia, T., et al. 1996. Biochim. Biophys.1293, 259.
Sang, Q.X., et al. 1995. Biochim. Biophys.1251, 99.
Zempo, N., et al. 1994. J. Vasc. Surg.20, 209.
Birkedal-Hansen, H. 1993. J. Periodontol64, 474.
Stetler-Stevenson, W.G., et al. 1993. FASEB J.7, 1434.
Jeffrey, J.J. 1991. Semin. Perinatol.15, 118.
Liotta, L.A., et al. 1991. Cell64, 327.
Harris, E. 1990. N. Engl. J. Med.322, 1277.
Legal Information
Storage Class
10 - Combustible liquids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico