94517
Quinoline
analytical standard
Synonyma:
1-Benzazine, 2,3-Benzopyridine
About This Item
Doporučené produkty
grade
analytical standard
Quality Level
vapor density
4.5 (vs air)
vapor pressure
0.07 mmHg ( 20 °C)
assay
≥98.0% (GC)
97.5-102.2% (wt., NT)
autoignition temp.
896 °F
shelf life
limited shelf life, expiry date on the label
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
refractive index
n20/D 1.625 (lit.)
bp
113-114 °C/11 mmHg (lit.)
237 °C (lit.)
mp
−17-−13 °C (lit.)
density
1.093 g/mL at 25 °C (lit.)
application(s)
cleaning products
cosmetics
flavors and fragrances
food and beverages
personal care
format
neat
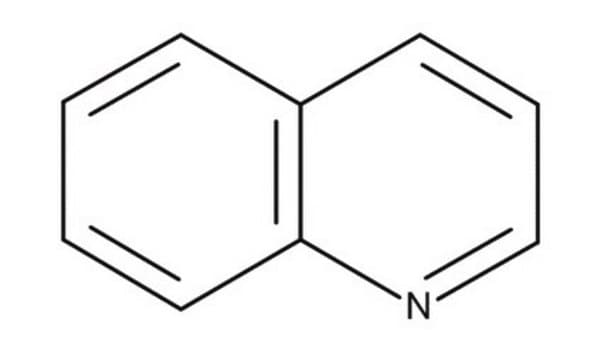
SMILES string
c1ccc2ncccc2c1
InChI
1S/C9H7N/c1-2-6-9-8(4-1)5-3-7-10-9/h1-7H
InChI key
SMWDFEZZVXVKRB-UHFFFAOYSA-N
Hledáte podobné produkty? Navštivte Průvodce porovnáváním produktů
Application
Recommended products
signalword
Danger
Hazard Classifications
Acute Tox. 3 Oral - Acute Tox. 4 Dermal - Aquatic Chronic 2 - Carc. 1B - Eye Irrit. 2 - Muta. 2 - Skin Irrit. 2
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 2
flash_point_f
213.8 °F - closed cup
flash_point_c
101 °C - closed cup
Vyberte jednu z posledních verzí:
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Zákazníci si také prohlíželi
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.