ABN185
Anti-alpha B-crystallin Antibody
from rabbit, purified by affinity chromatography
Synonyma:
Alpha-crystallin B chain, Alpha(B)-crystallin, Heat shock protein beta-5, Renal carcinoma antigen NY-REN-27, Rosenthal fiber component
About This Item
Doporučené produkty
biological source
rabbit
Quality Level
antibody form
affinity isolated antibody
antibody product type
primary antibodies
clone
polyclonal
purified by
affinity chromatography
species reactivity
bovine, human, rat
species reactivity (predicted by homology)
horse (based on 100% sequence homology), rhesus macaque (based on 100% sequence homology), ox (based on 100% sequence homology), mouse (based on 100% sequence homology)
technique(s)
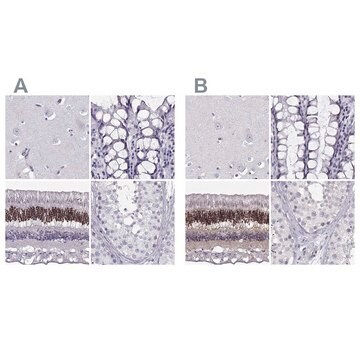
immunohistochemistry: suitable (paraffin)
western blot: suitable
NCBI accession no.
UniProt accession no.
shipped in
wet ice
target post-translational modification
unmodified
Gene Information
bovine ... Cryab(281719)
human ... CRYAB(1410)
mouse ... Cryab(12955)
rat ... Cryab(25420)
General description
Specificity
Immunogen
Application
Western Blot analysis: 0.05 µg/mL of this antibody detected Alpha-crystallin B chain in 10 µg of human kidney and calf kidney tissue lysate.
Neuroscience
Developmental Neuroscience
Quality
Western Blot analysis: 0.05 µg/mL of this antibody detected Alpha-crystallin B chain in 10 µg of bovine calf lens tissue lysate.
Target description
Physical form
Storage and Stability
Analysis Note
Bovine calf lens, human kidney and calf kidney tissue lysate
Other Notes
Disclaimer
Ještě jste nenalezli správný produkt?
Vyzkoušejte náš produkt Nástroj pro výběr produktů.
Storage Class
12 - Non Combustible Liquids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Osvědčení o analýze (COA)
Vyhledejte osvědčení Osvědčení o analýze (COA) zadáním čísla šarže/dávky těchto produktů. Čísla šarže a dávky lze nalézt na štítku produktu za slovy „Lot“ nebo „Batch“.
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.