678937
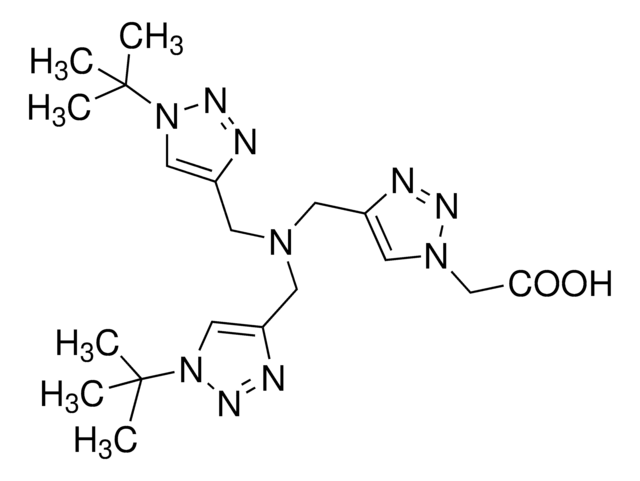
Tris[(1-benzyl-1H-1, 2, 3-triazol-4-yl)methyl]amine
97%
Synonyme(s) :
TBTA
About This Item
Produits recommandés
Pureté
97%
Forme
solid
Pertinence de la réaction
reagent type: ligand
reaction type: click chemistry
Pf
132-143 °C
Température de stockage
−20°C
Chaîne SMILES
C(N(Cc1cn(Cc2ccccc2)nn1)Cc3cn(Cc4ccccc4)nn3)c5cn(Cc6ccccc6)nn5
InChI
1S/C30H30N10/c1-4-10-25(11-5-1)16-38-22-28(31-34-38)19-37(20-29-23-39(35-32-29)17-26-12-6-2-7-13-26)21-30-24-40(36-33-30)18-27-14-8-3-9-15-27/h1-15,22-24H,16-21H2
Clé InChI
WKGZJBVXZWCZQC-UHFFFAOYSA-N
Application
Produit(s) apparenté(s)
Mentions de danger
Classification des risques
Aquatic Chronic 4
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
Click chemistry is an approach to the synthesis of drug-like molecules that can accelerate the drug discovery process by using a few practical and reliable reactions.
Click chemistry is an approach to the synthesis of drug-like molecules that can accelerate the drug discovery process by using a few practical and reliable reactions.
Click chemistry is an approach to the synthesis of drug-like molecules that can accelerate the drug discovery process by using a few practical and reliable reactions.
Click chemistry is an approach to the synthesis of drug-like molecules that can accelerate the drug discovery process by using a few practical and reliable reactions.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique