128775
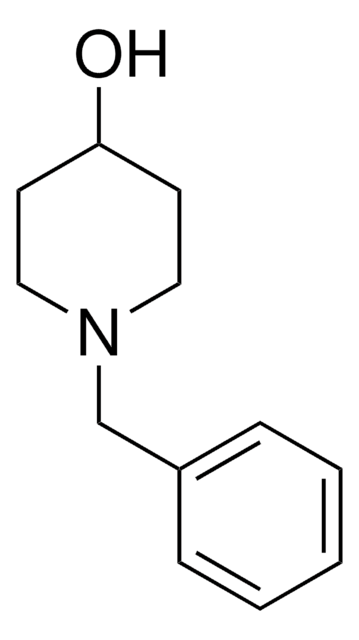
4-Hydroxypiperidine
98%
Synonym(s):
4-Piperidinol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H11NO
CAS Number:
Molecular Weight:
101.15
Beilstein:
102738
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
bp
108-114 °C/10 mmHg (lit.)
SMILES string
OC1CCNCC1
InChI
1S/C5H11NO/c7-5-1-3-6-4-2-5/h5-7H,1-4H2
InChI key
HDOWRFHMPULYOA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
The molecular structure, vibrational spectra, NBO and UV-spectral analysis of 4-Hydroxypiperidine have been studied. The compounds with a substituted 4-piperidinol core have been found to be potent antagonists of the human H(3) receptor.
Application
4-Hydroxypiperidine (4-Piperidinol) can be used in the synthesis of a highly potent and selective IP (PGI(2) receptor) agonist. It can also be used in the study of copper-catalyzed N- versus O-arylation.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
N- versus O-arylation of aminoalcohols: orthogonal selectivity in copper-based catalysts.
Alexandr Shafir et al.
Journal of the American Chemical Society, 129(12), 3490-3491 (2007-03-08)
James T Anderson et al.
Bioorganic & medicinal chemistry letters, 20(21), 6246-6249 (2010-09-14)
Compounds containing a substituted 4-piperidinol core have been found to be potent antagonists of the human H(3) receptor. The compounds exhibited up to a 60-fold preference for inhibiting the human H(3) receptor over the mouse and showed a low binding
Yasuhiro Morita et al.
Molecules (Basel, Switzerland), 17(2), 1233-1246 (2012-02-02)
An efficient synthesis of a highly potent and selective IP (PGI(2) receptor) agonist that is not structurally analogous to PGI(2) is described. This synthesis is accomplished through the following key steps: Nucleophilic ring-opening of 3-(4-chlorophenyl)-oxazolidin-2-one prepared by a one-pot procedure
S Sebastian et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 75(3), 941-952 (2010-01-09)
In this work, we report a combined experimental and theoretical study on molecular structure, vibrational spectra, NBO and UV-spectral analysis of 4-Hydroxypiperidine (4-HP). The FT-IR solid phase (4000-400 cm(-1)), FT-IR gas phase (5000-400 cm(-1)) and FT-Raman spectra (3500-50 cm(-1)) of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service