521396
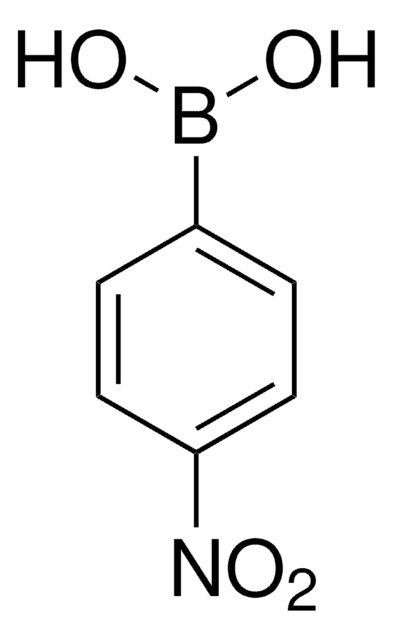
2-Cyanophenylboronic acid
≥95.0%
Synonym(s):
2-Cyanobenzeneboronic acid, [m-Cyanophenyl-4-yl]boronic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
NCC6H4B(OH)2
CAS Number:
Molecular Weight:
146.94
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
≥95.0%
mp
240 °C (dec.) (lit.)
functional group
nitrile
SMILES string
OB(O)c1ccccc1C#N
InChI
1S/C7H6BNO2/c9-5-6-3-1-2-4-7(6)8(10)11/h1-4,10-11H
InChI key
NPLZNDDFVCGRAG-UHFFFAOYSA-N
Related Categories
Application
Employed in a rhodium-catalyzed [3+2] annulation with alkynes leading to substituted indenones.
Useful in the synthesis of substitutes indenones or indanones
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Tomoya Miura et al.
Organic letters, 7(15), 3339-3341 (2005-07-16)
[reaction: see text]. A new [3 + 2] annulation reaction was developed in which 2-cyanophenylboronic acid reacted as a three-carbon component with alkynes or alkenes to afford substituted indenones or indanones. The use of an alkynoate even produced benzotropone, a
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service