319732
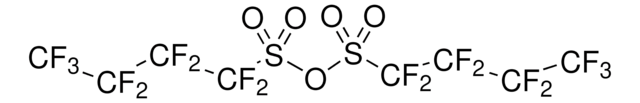
Perfluoro-1-butanesulfonyl fluoride
96%
Synonym(s):
NfF, Nonafluorobutanesulfonyl fluoride
Select a Size
About This Item
Recommended Products
Quality Level
assay
96%
form
liquid
reaction suitability
reaction type: click chemistry
refractive index
n20/D 1.3 (lit.)
bp
64 °C (lit.)
density
1.682 g/mL at 25 °C (lit.)
functional group
fluoro
SMILES string
FC(F)(F)C(F)(F)C(F)(F)C(F)(F)S(F)(=O)=O
InChI
1S/C4F10O2S/c5-1(6,3(9,10)11)2(7,8)4(12,13)17(14,15)16
InChI key
LUYQYZLEHLTPBH-UHFFFAOYSA-N
Application
NfF can also be used to prepare aryl nonaflates by reacting with corresponding aryloxysilanes in the presence of fluoride ion.[6]
related product
signalword
Danger
hcodes
Hazard Classifications
Skin Corr. 1B
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Faceshields, Gloves, Goggles
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)


![1-Chloromethyl-4-fluoro-1,4-diazoniabicyclo[2.2.2]octane bis(tetrafluoroborate) >95% in F+ active](/deepweb/assets/sigmaaldrich/product/structures/206/487/53d52ee5-ef71-4e9a-9bc8-938b68b98d5d/640/53d52ee5-ef71-4e9a-9bc8-938b68b98d5d.png)

![1,4-Diazabicyclo[2.2.2]octane ReagentPlus®, ≥99%](/deepweb/assets/sigmaaldrich/product/structures/366/129/a6ff4175-974d-4fac-9038-b35e508ef252/640/a6ff4175-974d-4fac-9038-b35e508ef252.png)