304484
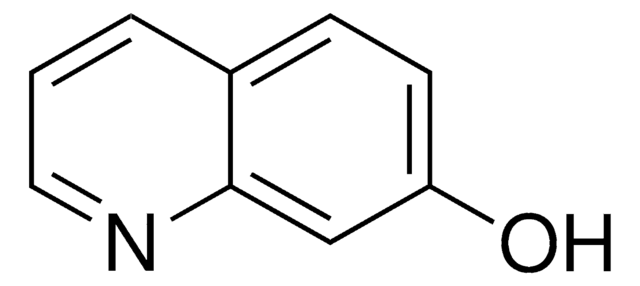
6-Hydroxyquinoline
95%
Synonym(s):
6-Quinolinol, 6-Hydroxyquinoline
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C9H7NO
CAS Number:
Molecular Weight:
145.16
Beilstein/REAXYS Number:
113196
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
95%
mp
188-190 °C (lit.)
SMILES string
Oc1ccc2ncccc2c1
InChI
1S/C9H7NO/c11-8-3-4-9-7(6-8)2-1-5-10-9/h1-6,11H
InChI key
OVYWMEWYEJLIER-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
6-Hydroxyquinoline is an ideal photoacid system for exploring excited-state proton transfer (ESPT) reactions. The excited-state proton transfer and geminate recombination of 6-hydroxyquinoline encaged in catalytic Na+-exchanged faujasite zeolites X and Y have been explored by measuring steady-state and picosecond time-resolved spectra.
Application
6-Hydroxyquinoline was used in synthesis of 2,6-substituted-benzo[d]thiazole analogs and 2,4-substituted-benzo[d]thiazole analogs.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Katharigatta Narayanaswamy Venugopala et al.
European journal of medicinal chemistry, 65, 295-303 (2013-06-04)
A novel and efficient one pot synthesis was developed for 2,6-substituted-benzo[d]thiazole analogues 4a-k and 2,4-substituted-benzo[d]thiazole analogues 4l-pvia three component condensation reaction of substituted arylaldehyde, 2-amino-6-halo/4-methyl-benzo[d]thiazole and 2-naphthol or 6-hydroxyquinoline in presence of 10% w/v NaCl in water by microwave method.
Yu-Hui Liu et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 128, 280-284 (2014-04-01)
6-Hydroxyquinoline (6HQ) is an ideal photoacid system for exploring excited-state proton transfer (ESPT) reactions. We have previously (Mahata et al. (2002)) shown that the ESPT reaction between 6HQ and trimethylamine (TMA) leads to an "unusual" emission in the 440-450 nm
Sun-Young Park et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 16(42), 12609-12615 (2010-09-21)
The excited-state proton transfer and geminate recombination of 6-hydroxyquinoline (6HQ) encaged in catalytic Na(+)-exchanged faujasite zeolites X (NaX) and Y (NaY) have been explored by measuring steady-state and picosecond time-resolved spectra. The pathways and rate constants of proton transfer of
Mohan Singh Mehata
The journal of physical chemistry. B, 112(28), 8383-8386 (2008-06-20)
A hydrogen-bonded network formed between 6-hydroxyquinoline (6-HQ) and acetic acid (AcOH) has been characterized using a time-resolved fluorescence technique. In the bridged hydrogen-bonded complex of cis-6-HQ and AcOH, an excited-state reaction proceeds via proton transfer along the hydrogen bond, resulting
M S Mehata et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 59(3), 559-567 (2003-01-14)
Fluorescence characteristics of 6-hydroxyquinoline (6-HQ) have been studied at room temperature in Nafion(R) film by steady state and nano-second time-resolved fluorescence spectroscopy. The fluorescence spectrum exhibits single emission band corresponding to the protonated form of 6-HQ in this matrix. However
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service