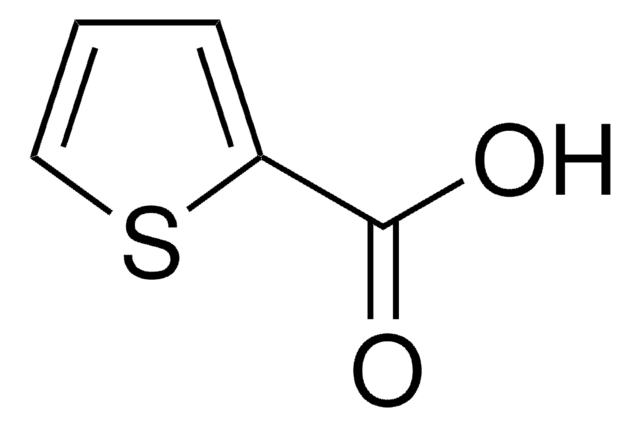
288985
2-Thiophenecarbonyl chloride
97%
Synonym(s):
α-Thenoyl chloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H3ClOS
CAS Number:
Molecular Weight:
146.59
Beilstein/REAXYS Number:
110145
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
97%
form
liquid
refractive index
n20/D 1.590 (lit.)
bp
206-208 °C (lit.)
density
1.371 g/mL at 25 °C (lit.)
functional group
acyl chloride
storage temp.
2-8°C
SMILES string
ClC(=O)c1cccs1
InChI
1S/C5H3ClOS/c6-5(7)4-2-1-3-8-4/h1-3H
InChI key
QIQITDHWZYEEPA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
The postfunctionalization reaction of 2-thiophenecarbonyl chloride with single walled carbon nanotubes (SWCNTs) was studied.
Application
2-Thiophenecarbonyl chloride was used in the synthesis of building blocks derived from ornithine by undergoing acylation/sulphonation of copper complex of orthinine.
signalword
Danger
hcodes
Hazard Classifications
Eye Dam. 1 - Met. Corr. 1 - Skin Corr. 1B
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 3
flash_point_f
194.0 °F
flash_point_c
90 °C
ppe
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Ferdinand Hof et al.
Journal of the American Chemical Society, 135(49), 18385-18395 (2013-11-22)
The reactivity of reduced single walled carbon nanotubes (SWCNTs) (carbon nanotubides), prepared under strict inert conditions in a glovebox with respect to the covalent functionalization with hexyl iodide and subsequent exposure to ambient conditions (air, moisture), was systematically investigated by
T Lescrinier et al.
The journal of peptide research : official journal of the American Peptide Society, 49(2), 183-189 (1997-02-01)
Recently great interest has arisen in the synthesis of combinatorial libraries, and this technology provides a significant partner to contemporary strategies in rational design and lead discovery. By simple combination of a given set of building blocks, high numbers of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![1,4-Diazabicyclo[2.2.2]octane ReagentPlus®, ≥99%](/deepweb/assets/sigmaaldrich/product/structures/366/129/a6ff4175-974d-4fac-9038-b35e508ef252/640/a6ff4175-974d-4fac-9038-b35e508ef252.png)