18565
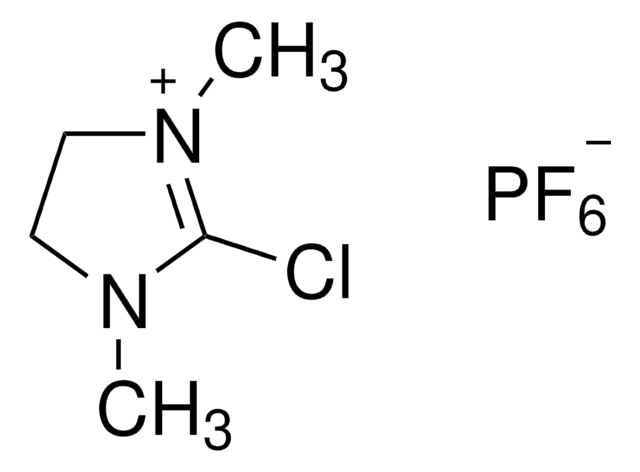
Bromotripyrrolidinophosphonium hexafluorophosphate
≥95.0% (HPLC), for peptide synthesis
Synonym(s):
PyBroP®
About This Item
Recommended Products
product name
Bromotripyrrolidinophosphonium hexafluorophosphate, ≥95.0% (HPLC)
Quality Level
assay
≥95.0% (HPLC)
form
solid
reaction suitability
reaction type: Coupling Reactions
application(s)
peptide synthesis
storage temp.
−20°C
SMILES string
F[P-](F)(F)(F)(F)F.Br[P+](N1CCCC1)(N2CCCC2)N3CCCC3
InChI
1S/C12H24BrN3P.F6P/c13-17(14-7-1-2-8-14,15-9-3-4-10-15)16-11-5-6-12-16;1-7(2,3,4,5)6/h1-12H2;/q+1;-1
InChI key
CYKRMWNZYOIJCH-UHFFFAOYSA-N
Application
Synthesis of primary amides
Direct dehydrative phosphonium cross coupling
Direct arylation
Pyrrolidide formation by phosphonium salt coupling reagents
- For Suzuki–Miyaura cross-coupling of phenols and arylboronic acids to synthesize biaryls and heterobiaryls.
- To functionalize pyrimidines (synthesized from 3,4-dihydropyrimidin-2(1H)-ones (DHPMs) via the Kappe dehydrogenation) by using various nucleophiles.
- To synthesize formamidines by coupling with various primary amines and N,N-diisopropylethylamine.
It can also be used as an activating reagent:
- For the activation of C-OH bond in tautomerizable heterocycles to form the phosphonium salt which aids the Sonogashira coupling of heterocycles with various alkynes.
- For the one–pot activation of C-O bond in phenols and further coupling reaction with phosphine oxide or phosphite to form C-P bonds.
Other Notes
Legal Information
signalword
Danger
hcodes
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service